Metabolism of Trans Polyunsaturated Fatty Acids Formed during Frying
The Authors: Jean Louis Sébédio and William W. Christie.
1. Introduction
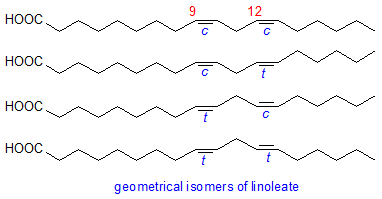
Double bonds in the fatty acids of vegetable oils subjected to the high temperatures during refining, especially the deodorisation process, and during frying can undergo geometrical isomerization from the cis to trans configuration. Oleic acid is hardly affected, while α-linolenic acid has a greater tendency to isomerization than linoleic acid. The two possible mono-trans isomers of linoleic (i.e. 9c,12t-18:2 and 9t,12c-18:2) are formed in roughly equal amounts, while the all-trans isomer (9t,12t-18:2) is produced at much lower concentrations.
Although eight geometric isomers are possible for α-linolenic acid, only four are detected under normal refining or frying conditions. These have been characterised as 9c,12c,15t-, 9c,12t,15c-, 9t,12c,15c- and 9t,12c,15t-18:3. Similarly, geometrical isomers of eicosapentaenoic and docosahexaenoic acids are formed during the refining of fish oils. However, if the oils have been subjected to partial hydrogenation prior to use in frying, they may contain a much wider range of trans-polyenes, including isomers with conjugated double bond systems and others with the double bonds separated by several methylene groups.
In this brief and subjective review, only those isomers formed by the heat treatments encountered during refining and frying are considered in detail. When consumed, these compete with the natural all-cis isomers for metabolism and indeed could interfere with the normal metabolism of polyunsaturated fatty acids with potentially harmful effects. The topic has been reviewed elsewhere by Emken [1], Sébédio and Chardigny [2], and Sébédio and Christie [3]. Here, only a few key references are listed (with apologies to other authors), and these review articles should be consulted for more detailed information including comprehensive reference citations.
2. Metabolism of trans Isomers in Vivo
Absorption and incorporation into tissues
Dietary triacylglycerols make up about 40% of the energy intake in the human diet in Western countries, and after ingestion most of the digestion is accomplished in the stomach by the reaction with pancreatic lipase. Fatty acids are released in free form from the 1(3) positions of the triacylglycerols with formation of 2-monoacyl-sn-glycerols. Both products are rapidly taken up by the intestinal cells, and are re-esterified into triacylglycerols before they are transported to the liver in the form of chylomicrons. In general, the experimental evidence is that most isomeric cis and trans di- and trienoic C18 fatty acids, when fed as part of a mixed diet, are as efficiently absorbed and incorporated into chylomicrons as the all-cis isomers.
When the chylomicrons reach the liver, the fatty acids are repackaged into triacylglycerols and phosphatidylcholine and thence into very-low-density and low-density lipoproteins, where dietary trans fatty acids may have an effect on cholesterol concentrations and thence on the risk of coronary heart disease [1]. Although the relative rates of incorporation may not differ, there is specificity in the manner in which trans fatty acids are utilized by acyltransferases. Thus di-trans 18:2 was incorporated preferentially into position 1 of phosphatidylcholine and into the sn-1 and sn-3 positions of the triacylglycerols, like the saturated fatty acids. In contrast, 9c,12t-18:2, like linoleic acid per se, was incorporated into the sn-2 position. Subsequently in human plasma, the enzyme lecithin:cholesterol acyl transferase discriminates strongly against the incorporation of trans isomers of linolenic acid from phosphatidylcholine into cholesterol esters [4].
Once the plasma lipids reach other tissues, both cis and trans fatty acids are rapidly taken up and incorporated into tissue lipids. However, 9c,12c,15t-18:3 is selectively incorporated into cardiolipin in a very similar manner to linoleic acid, its structural analogue. It therefore appears that the trans-15 ethylenic bond may be perceived as a single bond by the acyltransferases involved [5]. Support for this hypothesis comes from a finding that esterification of 9c,12t-18:2 into position 2 of phosphatidylcholine was similar to that of 9c-18:1.
Desaturation and elongation
As long ago as 1963, it was demonstrated that 9-cis,12-trans-linoleate was incorporated into an arachidonate analogue, but it was much later before this was confirmed to be 5-cis,8-cis,11-cis,14-trans-20:4 isomer.
![]()
Studies with mice fed isotopically labelled 18:2 isomers (9c,12c, 9c,12t and 9t,12c) showed that C20 metabolites were formed from all, and they were incorporated into many different tissues with a similar pattern in plasma and in liver lipids [6]. However, the all-cis metabolites were much more effectively utilized than those derived from 9c,12t-18:2. In contrast, the 20:4 metabolite of 9c,12t-18:2 was esterified more than twice as readily as that derived from linoleate per se. Most studies suggest that dietary di-trans-18:2 is not metabolized in this way, although it can be transformed to the expected 20:3 (but not 20:4) isomer when injected intracranially into rat brain.
Similarly, some trans-8:3 isomers can also be desaturated and elongated into trans isomers of eicosapentaenoic and docosahexaenoic acids in vivo. In particular in feeding experiments with rats, 9t,12c,15c-18:3, 9c,12c,15t-18:3 and to a lesser extent 9t,12c,15t-18:3 were elongated and desaturated to the expected 20:5 and 22:6 isomers. However, only the 17-trans isomer of 20:5 was incorporated into the neutral lipids and most of the phospholipid classes to a significant extent in tissues such as the liver, heart, kidneys, adrenals and testes. However, greater incorporation of trans-20:5 and 22:6 isomers was observed in brain and retina, but in a selective and time-dependent manner [7]. The retina was found to be more susceptible to modification by dietary lipids than the brain.
Beta oxidation
Studies of the oxidative metabolism of linoleic and α-linolenic acids and of their mono-trans isomers demonstrated that these were oxidized to between 56% and 74% over a 24-hour period, with the 18:3 isomers being oxidized to a greater extent. Retention of 9c,12t-18:2 in the liver and heart was significantly lower than for the other 18:2 isomers, especially in comparison to linoleic acid. On the other hand, no major differences were found for the 18:3 geometrical isomers in the rate of oxidation or in the amount of incorporation into tissue lipids [8].
Effects on the metabolism of other fatty acids
The desaturation and elongation of di- and polyenoic fatty acids of the n-6 and n-3 families can potentially be inhibited by all unsaturated fatty acids by mechanisms that include substrate inhibition, product inhibition, and competitive inhibition. The position and configuration of double bonds in the inhibitor determine the relative extent and the nature of this process.
While pioneering work with dietary di-trans-18:2 was found to inhibit the elongation and desaturation of linoleic and linolenic acids in early experiments with rats in the laboratories of R.T. Holman and O.S. Privett, it was shown subsequently by others that this only occurred when very high levels were fed. Similarly, mono-trans 18:2 isomers in the diet did not affect the level of arachidonic acid in the liver when fed at the low levels likely to be encountered in normal diets.
On the other hand, trans-18:3 isomers fed to animals in the form of heat-isomerized linseed oil brought about a decrease in the concentration of 20:4(n-6) and in the ratio of 20:4(n-6) to 18:2(n-6) in the phospholipids of liver in comparison to when fresh linseed oil was fed. Increased desaturase activity was observed, however, when the animals were fed diets deficient in n-3 fatty acids, presumably because the trans-18:3 isomers made up for this deficiency in part [9].
3. Metabolism of Trans Isomers in Vitro
Absorption and incorporation into tissues
The metabolism of synthetic 14C-labelled all-cis and mono-trans-18:2 and 18:3-isomers has been studied in a perfused liver model in the rat, and major differences in the manner of esterification were observed [10]. For example, much higher concentrations of trans-18:2 isomers were incorporated into triacylglycerols than was observed for the all-cis isomer. The remainder was incorporated into phospholipids, with most in phosphatidylcholine, more readily than the trans-isomers and very little of the latter was found in phosphatidylinositol especially.
With the 18:3 isomers (9c,12c,15c-, 9c,12c,15t-, and 9t,12c,15c-), the 14C–label was partitioned equally between phospholipids and triacylglycerols in each case, though somewhat more of the 9c,12c,15t isomer was found in phosphatidylcholine. No differences were observed for phosphatidylinositol in this instance. It appears that these fatty acids are recognized in the same manner by the acyl-CoA:phospholipid acyltransferases.
Desaturation and elongation
The Δ6 desaturation of 18:2 isomers in vitro has been studied using various models, including liver microsomal preparations, cultured cells and the perfused liver. For example, the 9c,12t-18:2 isomer was a much better substrate for the desaturase in studies with microsomes than was the 9t,12c isomer. In contrast, 9t,12c-18:2 was better elongated than 9c,12c, although the latter was more efficiently metabolized overall [11]. At low concentrations, the mono-trans isomers only inhibited the Δ6 desaturation of the all-cis 18:2 isomer slightly. In similar experiments with cultured glioma cells, the presence of a Δ12 trans bond was found to inhibit the formation of both 20:4n-6 and 20:5n-3 [12]. In the perfused rat liver model, in comparison to linoleic acid, the trans geometry in the Δ12 position greatly increased the amount of desaturation of the precursor, while the trans geometry in the Δ9 position increased the rate of elongation.
For the 18:3 isomers in the perfused rat liver model, in comparison with linolenic acid, trans-geometry in the Δ15 position decreased the extent of desaturation only. trans-Geometry in the Δ9 position both decreased the desaturation and increased the elongation of the precursor into 20:3 end products, results which correlated well with findings in vivo. In experiments with bovine endothelial cells in vitro, it was observed that 20:5 isomers (11t-20:5, 17t-20:5, and 11t,17t-20:5) formed by desaturation and elongation of the different 18:3 geometrical isomers could all be incorporated into lipids and further elongated into trans-22:5 and trans-24:5 isomers [13].
Beta oxidation
There are separate β-oxidation systems in mitochondria and peroxisomes to degrade fatty acids in animals, but the mitochondrial system is the more important quantitatively, especially for the more conventional range of fatty acids. There have been comparatively few studies of oxidation of trans polyenes, but experiments with rat liver mitochondria demonstrated that 9c,12t-18:2 was oxidized more rapidly than the other two isomers (as was also found in vivo) [8,10]. There is no obvious explanation for this in view of findings that while the acyl-CoA synthase activity had more affinity for the mono-trans isomers compared to linoleic acid, the carnitine palmitoyl transferase I had the same affinity for all the cis/trans-18:2 CoA esters.
Effects on the metabolism of other fatty acids
While it is well known that linoleic acid strongly inhibits the desaturation and elongation of α-linolenic acid and vice versa, comparatively few studies have been carried out on the effects of trans-polyenes on these process in vitro. However, experiments with cultured glioma cells showed that a trans ethylenic bond at the Δ12 position in a dienoic acid competitor inhibited the conversion of 9c,12c-18:2 to 5c,8c,11c,14c-20:4 [12]. Further, It was demonstrated earlier that dietary 9c,12t-18:2 and 9t,12c-18:2 induced changes in the Δ6 desaturation of 9c,12c-18:2 by rat liver microsomes in vitro [14]. Similar experiments with liver microsomes from rats fed a fat-free diet showed that 9c,12t-18:2 was a more effective inhibitor than 9t,12c-18:2 [11].
4. Effects on Eicosanoid Production
The eicosanoids are derived primarily from arachidonic acid by the action of cyclooxygenases and lipoxygenases, and they have a wide range of functions in tissues at low levels, especially in relation to inflammation. Fatty acids in the diet with trans double bonds could potentially inhibit eicosanoid metabolism by reducing the availability of substrates or by inhibiting specific enzymes.
Early experiments with high levels of dietary di-trans-linoleate demonstrated effects on eicosanoid production, but the conditions were not applicable to real diets. However, under more realistic conditions, trans-dienoic isomers in hydrogenated fat inhibited prostacyclin release by endothelial cells in the presence of a high level of linoleic acid [15]. Similar findings have been reported in a number of studies with different model systems, examining various eicosanoids and enzymes. For example, 14-trans-20:4 inhibited the conversion of 20:4(n-6) to thromboxanes, and was itself converted into other eicosanoid metabolites [14], while 17-trans-20:5 and 19-trans-22:6 inhibited the 12-lipoxygenase and cyclooxygenase pathways, respectively [16].
5. Human Studies
Geometrical isomers of linoleic acid, especially mono-trans-18:2, have been isolated and characterized from a number of human tissues, where they are presumed to be of dietary origin. Mono-trans isomers of linolenic acid have only rarely been detected. In the only human intervention study with the latter, parameters connected to cardiovascular risk factors were measured and it appeared that the result was a more atherogenic profile [17]. It was also found that one of the trans 18:3 isomers present in the diet, 9c,12c,15t-18:3, was metabolized to 5c,8c,11c,14c,17t-20:5 but no further in humans.
References
- Emken, E. Metabolism of trans and cis fatty acid positional isomers compared to non-isomeric fatty acids. In: Trans Fats in Foods, pp. 59-95 (G.R. List, W.M.N. Ratnayake and D. Kritchevsky (eds.), AOCS Press, Champaign, IL) (2007).
- Sébédio, J.L. and Chardigny, J.M. Biochemistry of trans polyunsaturated fatty acids. In: Trans Fatty Acids in Human Nutrition, pp. 191-215 (J.L. Sébédio and W.W. Christie (eds.), Oily Press, Dundee) (1998).
- Sébédio, J.L. and Christie, W.W. Metabolism of trans fatty acid isomers. In: Trans Fatty Acids in Human Nutrition (2nd edition), pp. 163-194 (F. Destaillats, J.L. Sébédio, F. Dionisi and J.-M. Chardigny (eds.), Oily Press, Bridgwater) (2009).
- Sébédio, J.L., Vermunt, S.H.F., Chardigny, J.M., Beaufrère, B., Mensink, R.P., Armstrong, R.A., Christie, W.W., Niemela, J., Hénon, G. and Riemersma, R.A. The effect of dietary trans α-linolenic acid on plasma lipids and platelet fatty acid composition: the TransLinE study. Eur. J. Clin. Nutr., 54, 104-113 (2000).
- Wolff, R.L., Combe, N.A., Entressangles, B., Sébédio, J.L. and Grandgirard, A. Preferential incorporation of dietary cis-9,cis-12,trans-15 18:3 acid into rat cardiolipins. Biochim. Biophys. Acta, 1168, 285-291 (1993).
- Beyers, E.C. and Emken, E.A. Metabolites of cis,trans, and trans,cis isomers of linoleic acid in mice and incorporation into tissue lipids. Biochim. Biophys. Acta, 1082, 275-284 (1991).
- Grandgirard, A., Bourre, J.M., Julliard, F., Homayoun, P., Dumont, O., Piciotti, M. and Sébédio, J.L. Incorporation of trans long-chain n-3 polyunsaturated fatty acids in rat brain structures and retina. Lipids, 29, 251-258 (1994).
- Bretillon, L., Chardigny, J.M., Sébédio, J.L., Poullain, D., Noël, J.P. and Vatèle, J.M. Oxidative metabolism of [1-14C] mono-trans isomers of linoleic and α-linolenic acids in the rat. Biochim. Biophys. Acta, 1390, 207-214 (1998).
- Blond, J.P., Chardigny, J.M., Sébédio, J.L. and Grandgirard, A. Effects of dietary 18:3 n-3 trans isomers on the Δ6 desaturation of α-linolenic acid. J. Food Lipids, 2, 99-106 (1995).
- Bretillon, L., Chardigny, J.M., Noël, J.P. and Sébédio, J.L. Desaturation and chain elongation of [1-14C]mono-trans isomers of linoleic and α-linolenic acids in perfused rat liver. J. Lipid Res., 39, 2228-2236 (1998).
- Berdeaux, O., Blond, J.P., Bretillon, L., Chardigny, J.M., Mairot, T., Vatèle, J.M., Poullain, D. and Sébédio, J.L. In vitro desaturation or elongation of mono-trans isomers of linoleic acid by rat liver microsomes. Mol. Cell. Biochem., 185, 17-25 (1998).
- Cook, H.W. and Emken, E.A. Geometric and positional fatty acid isomers interact differently with desaturation and elongation of linoleic and linolenic acids in cultured glioma cells. Biochem. Cell Biol., 68, 653-660 (1990).
- Loï, C., Chardigny, J.M., Cordelet, C., Leclere, L., Genty, M., Ginies, C., Noël, J.P. and Sébédio, J.L. Incorporation and metabolism of trans 20:5 in endothelial cells. Effect on prostacyclin synthesis. Lipids, 35, 911-918 (2000).
- Berdeaux, O., Sébédio, J.L., Chardigny, J.M., Blond, J.P., Mairot, T., Vatèle, J.M., Poullain, D. and Noël, J.P. Effects of trans n-6 fatty acids on the fatty acid profile of tissues and liver microsomal desaturation in the rat. Grasas Aceitas, 47, 86-99 (1996).
- Kummerow, F.A., Mahfouz, M.M. and Zhou, Q. Trans fatty acids in partially hydrogenated soybean oil inhibit prostacyclin release by endothelial cells in presence of high level of linoleic acid. Prostaglandins Other Lipid Mediat., 84, 138-153 (2007).
- O'Keefe, S.F., Lagarde, M., Grandgirard, A. and Sébédio, J.L. Trans n-3 eicosapentaenoic and docosahexaenoic acid isomers exhibit different inhibitory effects on arachidonic acid metabolism in human platelets compared to the respective cis fatty acids. J. Lipid Res., 31, 1241-1246 (1990).
- Vermunt, S.H.F., Beaufrère, B., Riemersma, R.A., Sébédio, J.L., Chardigny, J.M. and Mensink, R.P. Dietary trans α-linolenic acid from deodorised rapeseed oil and plasma lipids and lipoproteins in healthy men: the TransLinE Study. Brit. J. Nutr., 85, 387-392 (2001).
In This Section
- Oil Refining
- Action of Natural Antioxidants During Frying
- Formation of New Compounds During Frying - General Observations
- Formation of cyclic fatty acids during frying
- Formation of Epoxy-, Keto- and Hydroxy-Fatty Acids
- Formation of Volatiles and Short-Chain Bound Compounds
- Formation of Dimers and Oligomers
- Oxysterol Formation Frying Oils
- Structural Analysis of the Cyclic Fatty Acids Formed during Frying
- Cyclic Fatty Acids: Isolation and Quantitative Analysis in Food and Biological Tissues
- Analysis of Used Frying Oils and Fats by High-Performance Size-Exclusion Chromatography
- Analysis of Trans Polyunsaturated Fatty Acids
- Determination of Polar Compounds in Used Frying Oils and Fats by Adsorption Chromatography
- Determination of Oxidized Monomeric, Dimeric and Oligomeric Triacylglycerols; Diacylglycerols and Free Fatty Acids
- Separation and Quantification of Oxidized Monomeric, Dimeric and Oligomeric Fatty Acids
- Analysis of Oxidized Fatty Acids
- Analysis of Oxidized Sterols in Frying Oils
- Metabolism and Physiological Effects of Cyclic Fatty Acids Formed from Linoleic and alpha-Linolenic Acids during Frying
- Metabolism and Physiological Effects of Cyclic Fatty Acids Formed from Linoleic and alpha-Linolenic Acids during Frying
- Metabolism of Trans Polyunsaturated Fatty Acids Formed during Frying
- Biological Effects of Frying Oils Mediated by the Activation of Peroxisome Proliferator-Activated Receptors (PPAR)
