Analysis of Trans Polyunsaturated Fatty Acids
The Author: Jean Louis Sébédio and William W. Christie
1. Introduction
Trans polyunsaturated fatty acids (PUFA) of C18, C20, and C22 chain lengths having up to six ethylenic bonds are part of the human diet. They are formed during heat treatments such as deodorization of vegetable or fish oils [1] or during frying operations [2]. This process tends to cause cis-trans isomerization of double bonds without changing their positions to a significant extent. Some trans polyunsaturated fatty acids are also from natural origins such as those containing conjugated double bonds or conjugated linoleic acid (CLA). This short review will only deal with trans polyunsaturated fatty acids having methylene-interrupted double bonds as information on CLA.
A nutritional intervention carried out on human has shown that, like the trans monounsaturated fatty acids, some of the trans-PUFA may have an effect on lipoprotein metabolism [3]. It is therefore very important to detect and quantify them in food products. This brief review will describe the two main approaches, gas-liquid chromatography and high-performance liquid chromatography (together with chemical degradative techniques) used to analyse trans polyunsaturated fatty acids in food products.
2. Gas-Liquid Chromatography
Isomers of linoleic and α-linolenic acids
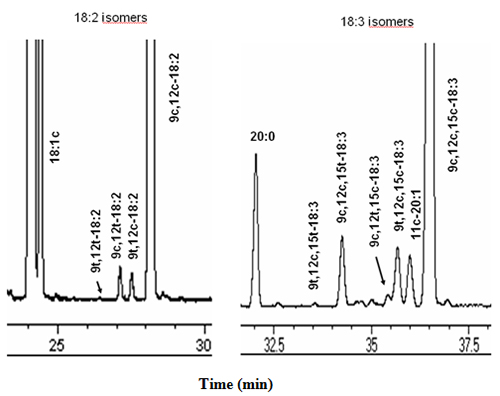
The three geometrical isomers of linoleic acid, namely 9t,12t-18:2, 9c,12t-18:2 and 9t,12c-18:2, which are very often present in refined, unhydrogenated liquid vegetable oils, are readily separated in the order stated on capillary columns coated with polar cyanosilicone. The four common geometric isomers of α-linolenic acid, i.e., 9t,12c,15t-18:3, 9c,12c,15t-18:3, 9c,12t,15c-18:3 and 9t,12c,15c-18:3, which are also very often present in refined liquid vegetable oils, give peaks that can be readily recognized in GC analyses on cyanosilicone capillary columns, and they are eluted from the column in order stated above (Fig. 1) [4,5].
Figure 1. The 18:2 (left) and 18:3 (right) regions of a GC chromatogram of FAMEs from a refined canola oil sample analysed using a SP-2560 capillary column (100 m × 0.25-mm I.D. × 20 μm film thickness), operated isothermally at 180°C. Hydrogen carrier gas, flow rate 1 mL/min.
The 18:2 isomer group of partially hydrogenated vegetable oils may contain up to 15 other 18:2 isomers. The identification of these is difficult, because suitable commercial standards are not available, except for 9t,12t-18:2, 9c,12t-18:2 and 9t,12c-18:2. However, these are the isomers most often encountered and their elution patterns have been established for SP-2560 and CP-Sil 88 columns. In general, such isomers elute in the order trans,trans < trans,cis < cis,trans followed by cis,cis. We will not extend discussion on this subject because of the efforts made by the industry to reduce the amount of partially hydrogenated oil in human diet. However, pertinent information has been published [6].
Even greater care must be taken when analysing the 18:3 isomers present in some refined vegetable oils. 11c-Eicosenoic acid (11c-20:1) is a natural monounsaturated fatty acid present in appreciable amounts in some vegetable oils, such as peanut oil and canola oil. Animal fats, especially lard, also contain this fatty acid but at lower levels. The 11c-20:1 elutes in the 18:3 region of the chromatogram and its relative retention time with respect to the 18:3 varies with the column temperature. Depending on the temperature of the column, it may elute before, with or after α-linolenic acid (9c,12c,15c-18:3). Therefore, an understanding of these variations is critical for the correct identifications of all the peaks in the 18:3 region of the chromatogram and for achieving correct fatty acid compositional data (for details see [5,7]).
Isomers of eicosapentaenoic and docosahexaenoic acids
Studies carried out on the development of analytical methods for identifying and quantifying trans polyunsaturated fatty acids with 5 or 6 ethylenic bonds are rather scarce and literature on the subject is limited [8-11]. In one study [8], trans isomers of eicosapentaenoic (EPA, 20:5n-3) and docosahexaenoic (DHA, 22:6n-3) acids were prepared by isomerization with p-toluenesulfinic acid and the isomers fractionated by silver ion HPLC in order to have pure fractions of mono- and di-trans isomers. BPX-70 columns seem to have a more suitable selectivity for the analysis of trans EPA and DHA isomers compared to SP-2560. Polyethylene glycol (PEG) phases may also be used to analyse trans isomers of long-chain PUFA in fish oil concentrates only if 22:0 and 22:1 are not present in significant amounts [9].
Two-dimensional fatty acid retention indices (for calculation see ref. 12) were found suitable and better than equivalent chain-length (ECL) values for identification of trans geometry in these polyunsaturated fatty acids [8].
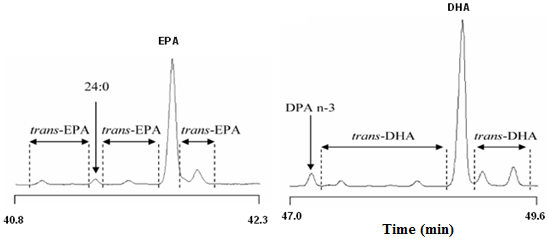
Quantification of geometrical isomers of highly unsaturated fatty acids such as EPA and DHA is a complex issue considering the number of components and isomers that may result from a heat treatment [10]. A validated method was recently published by Fournier et al. [11] to quantify low amounts of geometrical isomers of EPA and DHA in refined fish oils. It consists of converting the fish oil in to methyl esters and then analysing these by GC on a 100 m CP-Sil 88 column using a temperature programming method and hydrogen as the carrier gas. The elution of the EPA and DHA geometrical isomers present in a fish oil deodorized at 220°C is presented in Figure 2. The only drawback of such an analysis is the co-elution of a minor mono-trans isomer of DHA and all-cis DHA. However, for samples containing low levels of trans isomers this led to an underestimation of about 10%, which is within the magnitude of uncertainty of the method.
Figure 2. GC chromatogram of fish oil (deodorized at 220°C for 3 h) FAMEs. Zones of elution (left) of trans isomers of EPA and (right) of DHA. From Fournier et al. [11].
3. High-Performance Liquid Chromatography (HPLC)
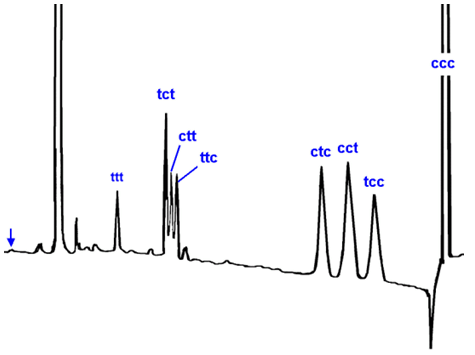
For definitive identification of trans polyunsaturated fatty acids, it is necessary that they are each isolated in sufficient amount and purity for chemical characterization. Although silver ion TLC is useful for obtaining a total trans-18:3 fraction from isomerized oils [13], HPLC in the silver ion mode appears to be the most useful micropreparative method for isolation of individual isomers, as illustrated in Figure 3 for α-linolenic acid [14].
Figure 3. Separation of phenacyl esters of geometrical isomers of α-linolenate by HPLC in the silver ion mode with UV detection at 238 nm. The mobile phase was a gradient of dichloromethane and methanol, the column (25 cm × 4.8 mm ID) was packed with Nucleosil 5SATMconverted to the silver ion form, and it was maintained at 10°C [14].
The all-trans isomer eluted first, followed by each of the di-trans isomers, then the mono-trans isomers and finally all-cis-18:3. Similar results were obtained by Adlof, who was also able to resolve 15 of the 16 possible cis/trans-isomers from arachidonate in the form of the methyl esters on a Chromspher LipidsTM column with hexane-acetonitrile (98.5:1.5, v/v) as mobile phase [15]. Later, Mjøs was able to separate methyl esters of isomerized EPA and DHA into groups according to the number of trans double bonds, although individual isomers were not resolved [8].
4. Position of a trans Double Bond in a Fatty Acid Chain
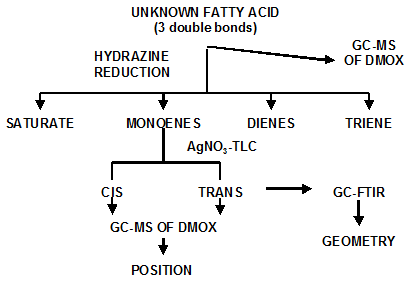
Determination of the position of a trans double bond on the carbon chain is a complex procedure (Fig. 4), which involves a combination of partial hydrogenation of the purified fatty acid, isolation of the resulting monoenes and separation into cis and trans fractions by silver nitrate thin-layer chromatography or HPLC, and subsequent analysis of each fraction by GC coupled with Fourier-transform infrared spectroscopy (GC-FTIR) and by GC coupled with mass spectrometry (GC-MS) of the dimethyloxazoline (DMOX) derivatives.
Figure 4. Procedure for determining double bond configurations in trans polyunsaturated fatty acids.
Partial hydrogenation of the unknown polyunsaturated fatty acid is a critical step, which is usually carried out using hydrazine reduction as described by Ratnayake et al. [16]. From a fatty acid having 3 double bonds, hydrazine reduction will give a mixture of the saturated fatty acid, 3 monoenes, 3 dienes and some unreacted fatty acid. As the hydrogenation takes place without modification of the position or the geometry of the original double bond, the double bonds in the three monoenes will be representative of the true positions. Consequently, structure elucidation is carried out on the monoenes. While the position of each ethylenic bond is given by GC-MS, the geometry of each double bond can be confirmed by GC-FTIR, although the elution characteristics on silver ion chromatography are usually a reliable guide.
References
- Ledoux, M., Juaneda, P., and Sébédio, J.L. Trans fatty acids: Definition and occurrence in foods. Eur. J. Lipid Sci. Technol., 109, 891-900 (2007).
- Sébédio, J.L. and Juanéda. P. Isomeric and cyclic fatty acids as a result of frying. In: Deep Frying, pp. 57-86 (M.D. Erickson (ed.), AOCS Press, Champaign) (2007).
- Vermunt, S.H.F., Beaufrère, B., Riemersma, R.A., Sébédio, J.L., Chardigny, J.M. and Mensink, R.P. Dietary trans α-linolenic acid from deodorised rapeseed oil and plasma lipids and lipoproteins in healthy men: the TransLinE Study. Brit. J. Nutr., 85, 387-392 (2001).
- Grandgirard, A., Sébédio, J.L. and Fleury, J. Geometrical isomerization of linolenic acid during heat treatment of vegetable oils. J. Am. Oil Chem. Soc., 61, 1563-1568 (1984).
- Sébédio, J.L. and Ratnayake, W.M.N. Analysis of trans mono- and polyunsaturated fatty acids. In: Trans Fatty Acids, pp. 102-131 (A.J. Dijkstra, R.J. Hamilton, and W. Hamm(eds.), Blackwell, Oxford) (2008).
- Ratnayake, W.M.N., Hansen, S.L. and Kennedy, M.P. Evaluation of the CP-Sil 88 and SP-2560 GC columns used in the recently approved AOCS Official Method Ce 1h-05: determination of cis-, trans-, saturated, monounsaturated, and polyunsaturated fatty acids in vegetable or non-ruminant animal oils and fats by capillary GLC method. J. Am. Oil Chem. Soc., 83, 475-488 (2006).
- Wolff, R.L. Recent applications of capillary gas-liquid chromatography to some difficult separations of unsaturated fatty acids. In: New Trends in Lipid and Lipoprotein Analysis, pp. 147-180 (J.L. Sébédio and E.G. Perkins (eds.), AOCS Press, Champaign, IL) (1995).
- Mjøs, S.A. Properties of trans isomers of eicosapentaenoic and docosahexaenoic acid methyl esters on cyanopropyl stationary phases. J. Chromatogr. A, 1100, 185-192 (2005).
- Mjøs, S.A. Retention behavior of trans isomers of eicosapentaenoic and docosahexaenoic acid methyl esters on a polyethylene glycol stationary phase. Eur. J. Lipid Sci. Technol., 110, 547-553 (2008).
- Fournier, V., Destaillats, F., Lambelet, P., Dionisi, F., Sébédio, J.L., and Berdeaux, O. Degradation products formed from long chain PUFA during deodorization of fish oil. Lipid Technol., 19, 9-11 (2007).
- Fournier, V., Destaillats, F., Hug, B., Golay, P.A., Joffre, F., Juanéda, P., Semon, E., Dionisi, F., Lambelet, P., Sébédio, J.L., and Berdeaux, O. Quantification of eicosapentaenoic and docosahexaenoic acid geometrical isomers formed during fish oil deodorization by gas-liquid chromatography. J. Chromatogr. A, 1154, 353-359 (2007).
- Mjøs, S.A. Two dimensional fatty acid retention indices. J. Chromatogr. A, 1061, 201-209 (2004).
- Ratnayake, W.M.N. and Cruz-Hernandez, C. Analysis of trans fatty acids of partially hydrogenated vegetable oils and dairy fats. In: Trans Fatty Acids in Human Nutrition (2nd edition), pp. 105-146 (F. Destaillats, J.L. Sébédio, F. Dionisi and J.-M. Chardigny (eds.), Oily Press, Bridgwater) (2009).
- Juanéda, P., Sébédio, J.L. and Christie, W.W. Complete separation of the geometrical isomers of linolenic acid by high performance liquid chromatography with a silver ion column. J. High Resolut. Chromatogr., 17, 321-324 (1994).
- Adlof, R.O. Separation of cis and trans fatty acid methyl esters by silver ion high performance liquid chromatography. J. Chromatogr. A, 659, 95-99 (1994).
- Ratnayake, W.M.N., Grossert, J.S. and Ackman, R.G. Studies on the mechanism of the hydrazine reduction reaction - applications to selected monoethylenic, diethylenic and triethylenic fatty acids of cis configurations. J. Am. Oil Chem. Soc., 67, 940-946 (1990).
In This Section
- Oil Refining
- Action of Natural Antioxidants During Frying
- Formation of New Compounds During Frying - General Observations
- Formation of cyclic fatty acids during frying
- Formation of Epoxy-, Keto- and Hydroxy-Fatty Acids
- Formation of Volatiles and Short-Chain Bound Compounds
- Formation of Dimers and Oligomers
- Oxysterol Formation Frying Oils
- Structural Analysis of the Cyclic Fatty Acids Formed during Frying
- Cyclic Fatty Acids: Isolation and Quantitative Analysis in Food and Biological Tissues
- Analysis of Used Frying Oils and Fats by High-Performance Size-Exclusion Chromatography
- Analysis of Trans Polyunsaturated Fatty Acids
- Determination of Polar Compounds in Used Frying Oils and Fats by Adsorption Chromatography
- Determination of Oxidized Monomeric, Dimeric and Oligomeric Triacylglycerols; Diacylglycerols and Free Fatty Acids
- Separation and Quantification of Oxidized Monomeric, Dimeric and Oligomeric Fatty Acids
- Analysis of Oxidized Fatty Acids
- Analysis of Oxidized Sterols in Frying Oils
- Metabolism and Physiological Effects of Cyclic Fatty Acids Formed from Linoleic and alpha-Linolenic Acids during Frying
- Metabolism and Physiological Effects of Cyclic Fatty Acids Formed from Linoleic and alpha-Linolenic Acids during Frying
- Metabolism of Trans Polyunsaturated Fatty Acids Formed during Frying
- Biological Effects of Frying Oils Mediated by the Activation of Peroxisome Proliferator-Activated Receptors (PPAR)