Rubber Biosynthesis in Plants
The Authors: Katrina Cornish and Joshua J. Blakeslee
Natural rubber is a strategically essential raw material throughout the world; global consumption is nearly 11 million metric tons each year, and demand is rapidly increasing. The United States is currently totally dependent upon the import of more than 1.2 million metric tons of natural rubber (NR) per year from tropically grown sources. The economic importance of maintaining a steady supply of NR is highlighted by the fact that the United States rubber products trade is worth over $18 billion annually, with a deficit growing to 72% in October 2010 (US Department of Commerce data). For more than a century, Hevea brasiliensis, or the para rubber tree, has provided almost all of the world's natural rubber, from a plantation and smallholding system involving tapping latex by hand. Most NR is produced in Southeast Asia, with <10% from Africa and ~1% from South America. Natural rubber prices reached all-time highs in 2011, primarily due to dramatically increased demand from both China and India. The independent International Rubber Study Group projects a 1.5 to 3 million metric ton shortfall in NR production by 2021. If the means to increase natural rubber production are not developed to meet rising NR demands, consumers worldwide will face a steep escalation in prices for products incorporating this material. While many nations are currently financing enormous expenditures to protect and develop both oil and mineral interests, it is surprising that means of augmenting the production of natural rubber, a vital strategic resource, are not being pursued.
The current market shortfall of NR is exacerbated by increasing petroleum costs driving up prices in the synthetic rubber industry, which is facing a shortage of butadiene, the monomer from which 80% of synthetic rubber is produced. Taken together, these issues prevent the natural and synthetic rubber markets from compensating for each other's high prices and/or production shortfalls, which they have historically been able to do. Furthermore, the genetic base of H. brasiliensis is extremely narrow, and vast acreages are planted as genetically identical clones, making the plantations prone to crop failure. South American Leaf Blight (SALB), the most deadly known H. brasiliensis disease caused by Microcyclus ulei, destroyed the South American rubber industry in the early twentieth century, with the result that very little rubber is produced in Brazil up to the present date. More recently, this devastating fungal pathogen has appeared in both India and Thailand and threatens the irreplaceable and economically vital populations of H. brasiliensis in these nations. Should SALB escape control measures, the current production of natural rubber in Southeast Asia could collapse with little or no advance warning.
The de facto selection of a single species to produce a globally important material has to date been largely a matter of chance and convenience. Natural rubber production is not limited to H. brasiliensis, however, and NR is produced by at least 2,500 plant species in four of the six major superorders of the dicotyledonous plants, as well as some fungi (Lactarius sp.). The distribution of rubber biosynthesis in the plant kingdom indicates that the ability to produce rubber evolved 100-200 million years ago, before or as the early dicotyledonous Angiosperms diverged from each other. Although all NR-producing species localize rubber production in the cytosolic component, individual species exhibit many differences in the details of their rubber biosynthesis, accumulation and compartmentalization.
Natural rubber, chemically, is a cis-1,4-polyisoprene polymer composed from isopentyl pyrophosphate monomers; usually with a C15 trans tail derived from the original initiating allylic pyrophosphate (a specific type of diphosphate). Natural rubber is an end product of one branch of the ubiquitous plant isoprenoid/terpenoid synthesis pathway. NR product quality is related, on the molecular level, to the polymer molecular weight, branching, gel content (insoluble, cross-linked rubber), and to the composition of the nonrubber components of the latex, and, on the cellular level, to the rubber particles in which the rubber polymers are compartmentalized. Species-specific differences in these parameters may be exploited using biotechnological approaches to produce different types of rubber, which may open up new product applications. Several other species are in commercial development, most notably Parthenium argentatum (guayule) and Taraxacum kok-saghyz (rubber, or Russian, dandelion), but Helianthius annuus, Chrysothamnus nauseosus, Cryptostegia grandifora, Solidago sp., Lactiflua sp. or Euphorbia lactiflua latex?, Ficus sp. and Euphorbia sp. and others have received focused attention.
Role of Rubber in Plants
In individual species, rubber-containing latex may play a role in disease resistance, wound healing or tolerance to environmental stress. Certainly, the cold-induction of rubber biosynthesis in P. argentatum is well established, as is the presence of antifungal and coagulant proteins in H. brasiliensis. However, remarkably, there is no unifying physiological role identified for rubber in plants. One possibility that has been raised is that rubber, an end product, serves, in part, to help condition plants to survive deleterious external stimuli. For example, rubber is produced in cold temperatures in temperate species like T. kok-saghyz and P. argentatum, and may play an important role in wound responses in tropical species such as H. brasiliensis and F. elastica. Additionally, in P. argentatum and H. brasiliensis (and possibly in other laticiferous species), no isoprene synthases are present to provide a means of respiring excess photosynthate (although larger volatile compounds are made) and rubber may serve as an excess assimilate sink to prevent damage to the photosynthetic apparatus under conditions of cold temperature and high light.
Rubber Particles
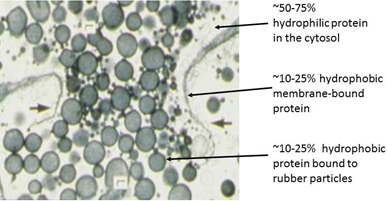
While relatively little is known about the physiological role of rubber synthesis in plant growth and development, the rubber biosynthetic particles (RPs), where the rubber polymerization reactions actually occur, have been more extensively studied. Rubber biosynthetic rate and polymer size are primarily regulated by the laticifers (Fig. 1), long multinucleate synciciate pipes formed from anatomized cells, in latex-producing species like H. brasiliensis, Ficus elastica, and T. kok-saghyz.
Figure 1. Transmission electronmicrograph of a rubber-containing laticifer (d'Auzac et al., 1989)
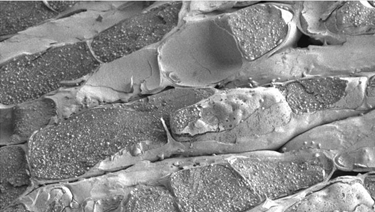
In nonlatex plants, such as P. argentatum, which produces rubber in bark parenchyma cells (Fig. 2) and transfers the mature rubber particles into the vacuole for storage, the regulation of rate and molecular weight has largely devolved to the rubber transferase enzyme itself. While studies to date have provided information on the size, composition, and synthetic capacity of developed, functional rubber particles, relatively little is known about the ontogeny, assembly and growth of RPs.
Figure 2. Scanning electron micrograph of bark parenchyma cells of stems of two-year old Parthenium argentatum, showing vacuoles filled with rubber particles (photograph by Delilah Wood, USDA).
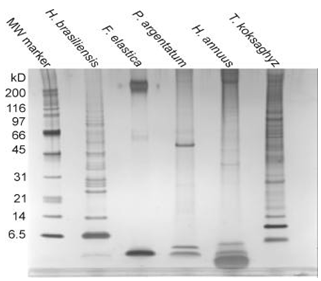
The rubber transferase protein, or protein complex, is embedded in a unilamellar biological membrane surrounding a rubber core, with new rubber molecules being synthesized from hydrophilic pyrophosphate substrates and elongated into the hydrophobic particle. The composition of the rubber particle monolayer membrane is species-specific, consisting of structural phospholipids and sterols, as well as a collection of proteins unique to each species (with very low sequence identity) (Fig. 3). The presence of glycosylated proteins in the rubber particle membrane suggests an origin in the rough endoplasmic reticulum and passage through the Golgi vesicular trafficking network, but this has yet to be proven. While phospholipid, sterol, and fatty acid (FA) compositions vary between H. brasiliensis, F. elastica, and T. kok-saghyz, all rubber particle membranes examined contain significant amounts of phosphatidic acid, which functions as both a structural phospholipid and a lipid signaling molecule.
Figure 3. SDA-PAGE gel of rubber particle associated proteins in different species (Dr. Wenshuang Xie).
As might be expected given the divergent protein profiles present in rubber particles from different species, many of the proteins present in these organelles do not share common three-dimensional structures. This lack of a conserved immune response-inducing epitope across RPs derived from varying species is why guayule latex products can be safely used by people with Type I latex allergic reactions induced by proteins present in H. brasiliensis-derived latex.
The properties of rubber produced in RPs are also impacted by the nonprotein membrane components present, which are highly species-specific. H. brasiliensis particles contain an unusual furanoid FA, whereas those from F. elastica contain very long chain FAs (Table 1).
| Table 1. Fatty acid compositions of the combined neutral, phospho- and glycolipids in rubber particles. | |||||||||||
| Species | 16:0 | 18:0 | 18:1 | 18:2 | 18:3 | 20:0 | 22:0 | 24:0 | 26:0 | 28:0 | Furanoid |
| Hevea | 4 | 10 | 4 | 11 | 71 | ||||||
| Guayule | 9 | 2 | 8 | 53 | 28 | ||||||
| Ficus | 10 | 16 | 4 | 21 | 18 | 13 | 18 | ||||
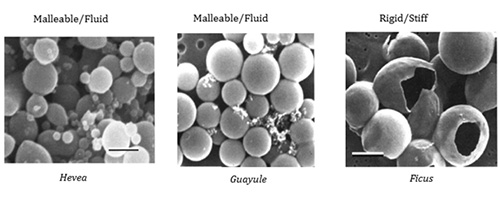
These F. elastica FAs, combined with the integral membrane proteins present in RPs in this species, lead to a stiff membrane, rather than the more common fluid membrane (Fig. 4).
Figure 4. Scanning electron micrographs of purified rubber particles (photograph by Delilah Wood, USDA).
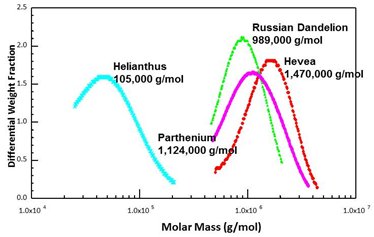
Rubber particles from different species all contain rubber cores, but not all rubber-producing species make high molecular weight rubber (Fig. 5). Helianthus annuus (sunflower), for example, makes mostly low molecular weight rubber. Molecular weight, however, is a prime determinant of rubber quality, with high quality being correlated high molecular weight. Also, not all rubber forms gel - the spontaneous cross-linking of insoluble polymer within the rubber particles - which can hamper processing. Gel does not seem to be related to the amount of protein per particle or the polymer molecular weight. Guayule makes high molecular weight polymer but with no gel content at all.
Figure 5. Size exclusion chromatograms of rubber purified from several species (Dr. Colleen McMahan)
Biochemical Regulation of Rubber Yield and Molecular Weight
Rubber biosynthesis is catalyzed by a membrane-bound cis-prenyl transferase, rubber transferase (EC 2.5.1.20), found at the surface of cytoplasmic rubber particles. Active rubber transferase has not been purified, reproducibly, to date, and so biochemical investigations have relied on using purified, enzymatically active rubber particles, from which all cytoplasmic components have been removed. This appears to be a valid approach because kinetic analysis indicates that only one isopentenyl pyrophosphate (IPP)-using enzyme is present when rubber is synthesized, which must be the rubber transferase. Thus, purified rubber particles present a single enzyme system, positioned on a suitable aqueous-organic interface, valid for biochemical investigation. Further, the study of biochemical parameters on the native particles is likely to provide kinetic information directly applicable to the in vivo situation, something always questionable in the study of solubilized membrane-bound enzymes.
Rubber transferases are a class of cis-prenyl transferases (CPTs) with substrate binding constants distinct from other soluble and membrane-bound CPTs and are able to synthesize varying molecular weight cis-1,4-polyisoprene, including above 1 million g/mole (Fig. 5).
Biochemical studies demonstrate that the regulation of biosynthetic rate (a direct component of yield), and mature rubber molecular weight (a prime component of rubber quality), are largely controlled by the concentration of initiator (an allylic pyrophosphate, the C15 trans-farnesyl pyrophosphate in vivo, although many allylic pyrophosphates of different size and stereochemistry are effective in vitro), monomer (the nonallylic, C5, isopentenyl pyrophosphate) and activator (divalent cation, magnesium in vivo, while manganese also is effective in vitro). The polymerization reaction catalyzed by rubber transferase is a condensation reaction, which releases a pyrophosphate and a proton, and specifically occurs via an alkylation by prenyl transfer to the initiating allylic pyrophosphate (APP) - electrophile – by the monomer isopentenyl pyrophosphate (IPP) – nucleophile. Thus, elongating rubber molecules are allylic pyrophosphates. Interestingly, different rubber-producing species have also evolved additional fine control mechanisms, and some rubber transferase enzymes are able to at least partially regulate the molecular weight of mature rubber polymers.
In vivo, rubber biosynthesis is limited by the supply of allylic pyrophosphate initiator, but even more so by the supply of IPP monomer. Recently, genetic transformation systems have been developed and engineered to “metabolically funnel” carbon into rubber production by both increasing the supply of IPP and initiating the synthesis of substrates that only rubber transferase can use. These systems are working well in P. argentatum and in T. kok-saghyz.
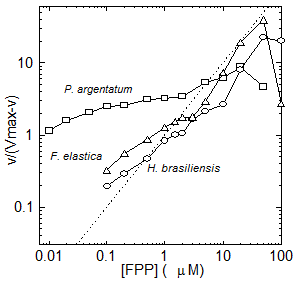
Extensive biochemical investigation of rubber biosynthesis in Hevea brasiliensis, Parthenium argentatum (guayule), Taraxacum kok-saghyz and Ficus sp. has demonstrated that plant species synthesize rubber in essentially the same way and their rubber transferases have similar characteristics – indeterminate product size, high KmIPP, high metal [A]max, metal cofactor requirements, and integral membrane localization. Further, in all species the concentration and reaction of monomer and initiator affect the biosynthetic rate, the number of rubber molecules synthesized and the molecular weight of the polymers produced. However, the divergent anatomical evolution in the above species has resulted in the co-evolution of several interesting differences in their respective rubber transferases, allowing all three to maintain rubber synthesis in their unique ecological niche. For example, P. argentatum (guayule) rubber transferase has a much wider range of negative cooperativity for the initiator farnesyl pyrophosphate (FPP) (Fig. 6) than is seen in the corresponding H. brasiliensis or F. elastica enzymes. In other words, when one FPP binds to the active site of guayule rubber transferase, the next FPP is unable to compete the first one off again to start a new rubber molecule. This, combined with the remarkably small KmFPP in this species, allows rubber transferase to effectively compete for FPP and IPP substrates and the plant to make high molecular weight rubber even in the summer time, when other enzymes compete for both of these monomers. In contrast, neither of the two laticiferous species, H. brasiliensis or F. elastica, shows this behavior, indicating that the laticifer regulates molecular weight in these species, rather than the rubber transferase itself.
Figure 6. Hill plot of IPP (v) kinetics plotted against log [FPP]. The linear dashed black line indicates Michaelis-Menten kinetics.
Similar to the control exerted by FPP binding (negative cooperativity), the initiation of rubber synthesis catalyzed by the rubber transferase complex also is regulated by the divalent cation magnesium. The differences in [A]maxMg2+, defined as the concentration of activator required for maximum velocity, observed in rubber transferase complexes correlate well with the actual endogenous Mg2+ concentrations in the latex of living plants. Consistent with these data, kinetic analyses have demonstrated that FPP-Mg2+ is the active substrate for rubber molecule initiation, although free FPP and Mg2+ can interact independently at the active site.
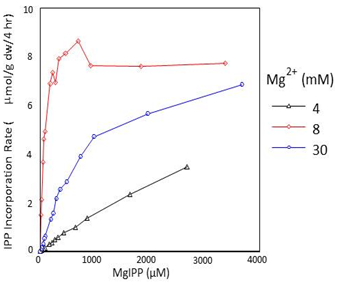
Once rubber synthesis has been initiated, Mg2+ can still interact independently at the active site with KdIPP-Mg < KdE-Mg. Unlike FPP, however, IPP alone is not able to interact independently with rubber transferases, and IPP-Mg is the only monomeric substrate utilized in the polymerization reactions responsible for rubber chain elongation. In vitro, the concentration of Mg2+ radically affects the affinity of the H. brasiliensis rubber transferase for IPP∙Mg (Fig. 7 and Table 2) and to a lesser extent in P. argentatum, further supporting a regulatory role for Mg2+ concentration in rubber chain elongation. The metal ion cofactor concentration also affects the molecular weight of the rubber produced by all three rubber-producing species.
Figure 7. Effect of IPP.Mg concentration on the IPP incorporation rate in Hevea brasiliensis purified, enzymatically-active rubber particles.
|
Table 2. Variation in KmIPP.Mg in H. brasiliensis and P. argentatum with Mg2+ concentration |
||||
| Hevea brasiliensis | Parthenium argentatum | |||
| Mg (mM) | KmIPP.Mg | KmIPP.Mg | ||
| SEa | SEa | |||
| μM | μM | |||
| 4 | 8000 | 600 | 1700 | 250 |
| 8 | 68 | 10 | 120 | 45 |
| 10 | - | - | 140 | 35 |
| 30 | 970 | 70 | - | - |
| 40 | - | - | 290 | 50 |
| a SE = standard error | ||||
The above data indicate that endogenous Mg2+ is likely very important for latex yield and quality. Thus, internal Mg2+ levels are an attractive parameter for targeted optimisation using agronomic procedures (i.e. adding magnesium fertilizer or a chelator) to maximize plant rubber production.
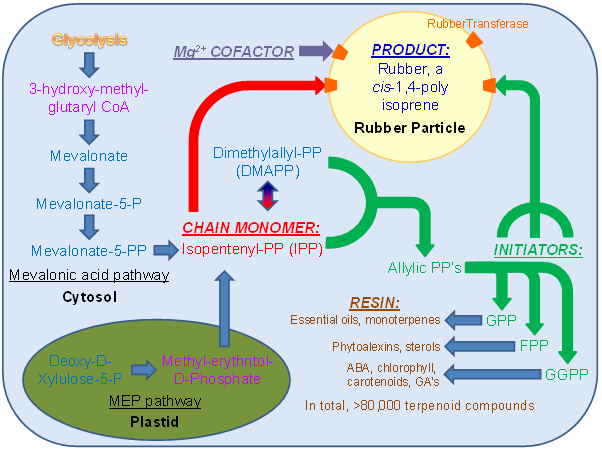
Figure 8. Metabolic map of rubber synthesis in plant cells. IPP (isopentenyl pyrophosphate), the rubber chain monomer essential for chain elongation (pathway in red) is synthesized via either the mevalonic acid pathway in the cytosol or the MEP pathway in the plastid (rate-limiting metabolic intermediates are shown in purple). IPP and its stereoisomer DMAPP are combined to make several allylic pyrophosphates. GPP (C-10), FPP (C-15), and GGPP (C-30) are able to serve as rubber chain initiators (pathway in green) in various species, and are also the building blocks for several other compounds found in the resin of rubber producing cells (pathway in brown). Rubber synthesis occurs in the unilamellar rubber particle (yellow), requires a Mg2+ ion cofactor (grey), and is mediated by the rubber transferase protein(s) (orange).
Proteins Involved in Rubber Biosynthesis
While various rubber particle bound proteins have been implicated in rubber biosynthesis, the lack of solubilized activity has made conclusive identification of a distinct “rubber transferase” protein or proteins very difficult. Candidate rubber biosynthetic proteins have been identified through their association with enzymatically active rubber particles and/or through temporal or spatial gene expression profiling correlated with rubber biosynthesis in specific plant tissues and organs. These methods have proven less than satisfactory, however, and further work has discredited two of the primary proteins identified in these studies: the 14.6 kDa so-called Rubber Elongation Factor (REF), which actually appears to have a role in latex coagulation (a defense mechanism); and the farnesyl pyrophosphate synthase, which was touted as undergoing a stereochemical shift to become the cis-prenyl transferase "rubber transferase", but which undergoes no such structural change.
Despite these drawbacks, genetic profiling approaches have given some insight into the peptide components of the rubber synthesis machinery. For example, the small rubber particle protein (SRPP) does appear to play a role in molecular weight regulation and possibly also in biosynthetic rate. SRPP, a 23 kDa acidic protein (pI = 4.8), was first identified in H. brasiliensis. Homologs have since been identified in other rubber-producing species, including P. argentatum and T. kok-sahgys. Several lines of evidence support a role for SRPP in rubber biosynthesis. Interestingly, recent studies also have determined that SRPP plays a role in latex coagulation.
In addition to molecular cloning, several other methods, including photo-affinity substrate labeling, are currently being used to identify proteins involved in rubber biosynthesis, although these data are, for the most part, not yet published. Interestingly, these studies have identified several cis-prenyl transferases isolated from the latex of H. brasilinesis and T. kok-saghyz which may be associated with rubber particles, which have been claimed to be rubber particle-bound rubber transferases. Unfortunately, however, none of these proteins has been proven to make high molecular weight polyisoprene and their kinetic constants do not match those of rubber transferase, itself.
Closing Comments
In conclusion, natural rubber is a vitally important strategic material, which is synthesized in diverse plant species. While production of rubber to date has focused on the tropical para rubber tree, Hevea brasiliensis, other species including Russian dandelion (T. kok-saghyz), guayule (P. argentatum), and ficus (Ficus sp.) also produce rubber, and the former two are currently being explored as alternate sources of natural production. While these organisms each exhibit some species-specific differences in the tissue localization and regulation of rubber production, the basic mechanism of rubber production is conserved across species, with the synthesis of this polyisoprenoid polymer being localized to cytosolic, unilamellar vesicles containing the protein or proteins making up the rubber transferase. In order to maximize rubber production from alternate rubber species, an increased understanding of the development, composition, and regulation of the protein(s) making up rubber transferases is essential. While molecular screening and reverse genetic approaches have provided some progress in this direction, ongoing studies utilizing photo-affinity labeled substrates and protein interactome studies promise to yield further advances in the field and help to ensure a steady production of natural rubber for the use of the growing global consumer market.
References
- Archer, B.L. and Audley, B.G. New aspects of rubber biosynthesis. Bot. J. Linn. Soc., 94, 181-196 (1987).
- Asawatreratanakul, K., Zhang, Y.W., Wititsuwannakul, D., Wititsuwannakul, R., Takahashi, S., Rattanapittayaporn, A. and Koyama, T. Molecular cloning, expression and characterization of cDNA encoding cis-prenyltransferases from Hevea brasiliensis. Eur. J. Biochem., 270, 4671-4680 (2003).
- Backhaus, R.A. and Walsh, S. The ontogeny of rubber formation in guayule, Parthenium argentatum Gray. Bot. Gaz., 144, 391-400 (1983).
- Benedict, C.R., Goss, R., Foster, M.A. and Greer, P.J. The formation of rubber particles in developing cortical parenchyma of Parthenium argentatum plants exposed to the low temperatures of fall and winter of the Chihuahuan Desert. Ind. Crop Prod., 30, 403-406 (2009) (DOI: 1016/j.indcrop.2009.07.009).
- Bowers, J.E. Natural rubber-producing plants for the United States. USDA, Cooperative State Research Service and the National Agricultural Library. National Agricultural Library, Beltsville, Maryland (1990).
- Castillón, J. and Cornish, K. Regulation of initiation and polymer molecular weight of cis-1,4-polyisoprene synthesized in vitro by particles isolated from Parthenium argentatum, Gray. Phytochemistry, 51, 43-51 (1999).
- Chow, K.S., Wan, K.L., Isa, M.N., Bahari, A., Tan, S.H., Harikrishna, K. and Yeang, H.Y. Insights into rubber biosynthesis from transcriptome analysis of Hevea brasiliensis latex. J. Exp. Bot., 58, 2429-2440 (2007).
- Cornish, K. The separate roles of plant cis and trans prenyl transferases in cis-1,4 polyisoprene biosynthesis. Eur. J. Biochem., 218, 267-271 (1993).
- Cornish, K. Similarities and differences in rubber biochemistry among plant species. Phytochemistry, 57, 1123-1134 (2001).
- Cornish, K. and Scott, D.J. Biochemical regulation of rubber biosynthesis in guayule (Parthenium argentatum Gray). Ind. Crop Prod., 22, 49-58 (2005) (DOI: 10.1016/j.indcrop.2004.04.032).
- Cornish, K., Wood, D.F. and Windle, J.J. Rubber particles from four different species, examined by transmission electron microscopy and electron-paramagnetic-resonance spin labeling, are found to consist of a homogeneous rubber core enclosed by a contiguous, monolayer biomembrane. Planta, 210, 85-96 (1999).
- da Costa, B.M.T., Keasling, J.D. and Cornish, K. Regulation of rubber biosynthetic rate and molecular weight in Hevea brasiliensis by metal cofactor. Biomacromolecules, 6, 279-289 (2005) (DOI: 10.1021/Bm049606w).
- d'Auzac, J., Jacob, J.L. and Chrestin, H. Physiology of Rubber Tree Latex. (CRC Press, Inc., 1989).
- Espy, S.C., Keasling, J.D., Castillon, J. and Cornish, K. Initiator-independent and initiator-dependent rubber biosynthesis in Ficus elastica. Arch. Biochem. Biophys., 448, 13-22 (2006) (DOI: 10.1016/j.abb.2006.01.009).
- Han, K.H., Shin, D.H., Yang, J., Kim, I.J., Oh, S.K. and Chow, K.S. Genes expressed in the latex of Hevea brasiliensis. Tree Physiol., 20, 503-510 (2000).
- International Rubber Study Group. Statistical Summary of World Rubber Situation (2010).
- Kang, H.S., Kim, Y.S. and Chung, G.C. Characterization of natural rubber biosynthesis in Ficus benghalensis. Plant Physiol. Biochem., 38, 979-987 (2000).
- Kush, A., Goyvaerts, E., Chye, M.L. and Chua, N.H. Laticifer-specific gene expression in Hevea brasiliensis (rubber tree). Proc. Natl. Acad. Sci. U.S.A., 87, 1787-1790 (1990).
- Oh, S.K., Kang, H., Shin, D.H., Yang, J., Chow, K.S., Yeang, H.Y., Wagner, B., Breiteneder, H. and Han, K.H. Isolation, characterization, and functional analysis of a novel cDNA clone encoding a small rubber particle protein from Hevea brasiliensis. J. Biol. Chem., 274, 17132-17138 (1999).
- Rao, B.S. Potential threat of South-American leaf blight to plantation rubber industry in Southeast-Asia and Pacific region. FAO Plant Protect. B, 21, 107-113 (1973).
- Rojruthai, P., Sakdapipanich, J.T., Takahashi, S., Hyegin, L., Noike, M., Koyama, T. and Tanaka, Y. In vitro synthesis of high molecular weight rubber by Hevea small rubber particles. J. Biosci. Bioeng., 109, 107-114 (2010).
- Salvucci, M.E., Barta, C., Byers, J.A. and Canarini, A. Photosynthesis and assimilate partitioning between carbohydrates and isoprenoid products in vegetatively active and dormant guayule: physiological and environmental constraints on rubber accumulation in a semiarid shrub. Physiol. Plant., 140, 368-379 (2010).
- Schmidt, T., Hillebrand, A., Wurbs, D., Wahler, D., Lenders, M., Schulze Gronover, C. and Prüfer, D. (2009) Molecular cloning and characterization of rubber biosynthetic genes from Taraxacum koksaghyz. Plant Mol. Biol. Rep., 28, 277-284 (2010) (DOI: 10.1007/s11105-009-0145-9).
- Scott, D.J., da Costa, B.M.T., Espy, S.C., Keasling, J.D. and Cornish, K. Activation and inhibition of rubber transferases by metal cofactors and pyrophosphate substrates. Phytochemistry, 64, 123-134 (2003) (DOI: 10.1016/S0031-9422(03)00266-8).
- Shimizu, N., Koyama, T. and Ogura, K. Molecular cloning of the undecaprenyl diphosphate synthase gene of Micrococcus luteus B-P 26. FASEB J., 11, A1247-A1247 (1997).
- Siler, D.J., Goodrich-Tanrikulu, M., Cornish, K., Stafford, A.E. and McKeon, T.A. Composition of rubber particles of Hevea brasiliensis, Parthenium argentatum, Ficus elastica, and Euphorbia lactiflua indicates unconventional surface structure. Plant Physiol. Biochem., 35, 881-889 (1997).
- Swanson, C.L., Buchanan, R.A. and Otey, F.H. Molecular-weights of natural rubbers from selected temperate zone plants. J. Appl. Polymer Sci., 23, 743-748 (1979).
- Wititsuwannakul, R., Rukseree, K., Kanokwiroon, K. and Wititsuwannakul, D. A rubber particle protein specific for Hevea latex lectin binding involved in latex coagulation. Phytochemistry, 69, 1111-1118 (2008) (DOI: 10.1016/j.phytochem.2007.12.007).
- Wood, D.F. and Cornish, K. Microstructure of purified rubber particles. Int. J. Plant Sci., 161, 435-445 (2000).
- Xie, W., McMahan, C.M., DeGraw, A.J., Distefano, M.D., Cornish, K., Whalen, M.C. and Shintani, D.K. Initiation of rubber biosynthesis: In vitro comparisons of benzophenone-modified diphosphate analogues in three rubber-producing species. Phytochemistry, 69, 2539-2545 (2008) (DOI: 10.1016/j.phytochem.2008.07.011).
- Yeang, H.Y., Cheong, K.F., Sunderasan, E., Hamzah, S., Chew, N.P., Hamid, S., Hamilton, R.G. and Cardosa, M.J. The 14.6 kd rubber elongation factor (Hev b1) and 24 kd (Hev b3) rubber particle proteins are recognized by IgE from patients with spina bifida and latex allergy. J. Allergy Clin. Immunol., 98, 628-639 (1996).
In This Section
- Plant Fatty Acid Synthesis
- Production of Unusual Fatty Acids in Plants
- Arabidopsis Acyl-Coenzyme A-Binding Proteins
- Long Chain acyl-coA Synthetases and Other Acyl Activating Enzymes
- Plant Triacylglycerol Synthesis
- Triacylglycerol Biosynthesis in Eukaryotic Microalgae
- Subcellular Oil Droplets and Oleosins in Plants
- Triacylglycerol Mobilisation in Plants
- Role of Transcription Factors in Storage Lipid Accumulation in Plants
- Biosynthesis of Plant Lipid Polyesters
- Rubber Biosynthesis in Plants
- Carotenoid Biosynthesis and Regulation in Plants
- The Oxylipin Biosynthetic Pathways in Plants
- N-Acylphosphatidylethanolamines (NAPEs), N-acylethanolamines (NAEs) and Other Acylamides: Metabolism, Occurrence and Functions in Plants
- Phosphoinositide Signaling in Plants
- Plant Lipidomics
- 50 years of Galactolipid Research: The Beginnings
- Transport and function of lipids in the plant phloem