Role of Transcription Factors in Storage Lipid Accumulation in Plants
The Author: Pierre R. Fobert National Research Council of Canada, 110 Gymnasium Place, Saskatoon, SK S7N 0W9, Canada
Introduction
With few exceptions, oil accumulates in seeds as triacylglycerols; three fatty acid molecules esterified to a glycerol backbone. The synthesis of normal fatty acids and unusual fatty acids, their assembly into triacylglycerols and subsequent packaging to form oil bodies have been reviewed elsewhere, and appear as separate contributions to the AOCS Lipid Library by Harwood, Hildebrand, Weselake et al., and Huang, respectively. These processes are highly regulated, involving spatial compartmentalization between different organelles and the exquisite control of several biosynthetic steps by one or more of a variety of biochemical mechanisms. The important role of transcriptional regulation towards seed oil accumulation has recently emerged. Not only are mRNA levels of many genes involved in seed oil biosynthesis coordinated during seed development, but altering the levels of certain transcription factors can affect the expression of multiple lipid biosynthetic genes, resulting in altered seed oil content.
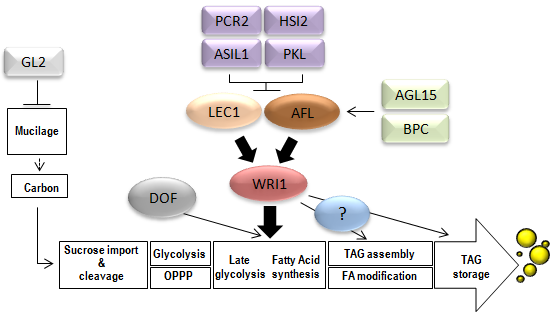
This article reviews salient features of plant transcription factors that have been implicated in the control of seed oil deposition. To set the stage, fundamental concepts in transcription factor biology are first summarized. Subsequently, the role of key transcription factors is highlighted by describing seed oil phenotypes associated with altering their expression levels by mutagenesis or transgenic means, and providing information on the putative target genes and pathways affected. The location of the transcription factors in hierarchical cascades and the modes of transcriptional regulation are also discussed, when this information is known. A summary of the information presented is depicted in Figure 1.
Figure 1. Transcription factors involved in seed oil synthesis. For simplicity, ABI3, LEC2 and FUS3 are shown as one entity (AFL). LEC1 and AFL control genes in most of the steps depicted and arrows are not shown for clarity. ‘?’ Represents an unknown factor proposed to be required to act with WRI1 in the regulation of genes involved in FA modification, TAG assembly and storage. PPP, pentose phosphate pathway; TAG, triacylglycerol; FA, fatty acid. Adapted from: Santos-Mendoza et al. Plant J., 54, 608-620 (2008) and Baud and Lepiniec, Prog. Lipid Res., 49, 235–249 (2010).
Transcription Factors 101
Transcription factors are sequence-specific DNA binding proteins that help recruit the transcriptional machinery to gene promoters. This typically involves conformational changes in chromatin structure to facilitate access to the DNA, although this aspect of gene regulation has not been well characterized for genes involved in seed oil synthesis. Plant genomes contain large numbers of genes capable of encoding transcription factors, which can be grouped into families based upon the structure of their DNA-binding domains. Some of the families with members implicated in regulating seed oil deposition are listed in Table 1. Proteins in different families recognize distinct, short (6-10 bp) stretches of DNA sequences with high affinity and, within families, subtle differences in DNA sequence binding preferences also exist. Gene promoters typically contain multiple cognate binding sites for one or more families of transcription factors, spread over several hundred to thousands of base pairs. Ultimately, transcription of a gene is determined by the combination of cis-acting cognate binding sites it contains and the repertoire of active transcription factors expressed in the cell.
| Table 1. Summary of transcription factors that have been implicated in the control of seed oil deposition. The list is not intended to be comprehensive. | ||
| Family | Transcription factor name | Summary of role in seed oil deposition |
| B3 domain; AFL Clade | ABSCISIC ACID INSENSITIVE3 (ABI3), LEAFY COTYLEDON2 (LEC2), FUSCA3 (FUS3) |
Master regulators of embryogenesis and seed maturation; mutation/overexpression often associated with pleiotropic effects; direct and indirect regulation of suites of genes involved in carbohydrate and lipid metabolism, including fatty acid synthesis, triacylglycerol assembly and packaging |
| HAP3/CBP | LEAFY COTYLEDON1 (LEC1), LEC1-LIKE (L1L) |
Subunits of CCAAT binding proteins; capable of working independently of CBP; master regulators of embryogenesis and seed maturation; direct and indirect regulation of genes involved in carbohydrate and lipid metabolism |
| AP2 | WRINKLED1 (WRI1) | Direct target of master regulators having more specific role towards seed oil biosynthesis; mutants dramatically reduced in seed oil content and wrinkled appearance; direct and indirect regulation of carbohydrate and lipid metabolism genes, particularly plastidial fatty acid synthesis |
| Dof | GmDof4 GmDof11 | Transgenic expression yields higher seed oil levels; direct and indirect regulation of lipid metabolism genes; possible negative regulators of seed storage proteins |
| CHD3 | PICKLE (PKL) | Putative chromatin remodeling factor; represses master regulator genes at germination; associated with the repressive chromatin mark H3K27me3 |
| PRC2 | FERTILIZATION INDEPENDENT ENDOSPERM (FIE), SWINGER (SWN), EMBRYONIC FLOWER2 (EMF2)< |
Components of Polycomb Repressive Complex 2 that catalyze deposition of H3K27me3; repressors of seed maturation genes in vegetative tissues |
| B3 domain; HSI2 Clade | HIGH-LEVEL EXPRESSION OF SUCROSE INDUCIBLE GENE2 (HSI2)/VAL1, HSI2-LIKE1 (HSIL1/VAL2), HSL2/VAL3 |
Act redundantly to repress AFL Clade genes and other positive regulators of seed maturation during germination and in seedlings; possible chromatin remodeling activities |
| AP2 | APETALA2 (AP2) | Negative regulator of seed size, possibly via carbohydrate metabolism in the seed coat; effects on seed oil deposition likely indirect |
| HD-ZIP | GLABRA2 (GL2) | Negative regulator of oil content; loss of seed mucilage proposed to make more C available for fatty acid synthesis |
Any given transcription factor may regulate the expression of one or more target genes, some of which may encode transcription factors themselves. These, in turn, may regulate the expression of other gene sets, yielding hierarchical transcriptional cascades. In theory, transcription factors near the top of the hierarchy may be responsible for controlling broad developmental and metabolic processes, involving multiple pathways, while those near the bottom may regulate specific biochemical steps within a pathway.
View from the Top: Master Regulators of Seed Maturation
The Arabidopsis transcription factors LEAFY COTYLEDON1 (LEC1), LEC2, ABSCISIC ACID INSENSITIVE3 (ABI3), and FUSCA3 (FUS3), and their homologues in other plant species, are generally considered as master regulators of embryogenesis and seed maturation, being situated at, or near, the top of transcriptional cascades. Mutation of the genes encoding these transcription factors can have severe detrimental effects on seed maturation, including reductions in seed oil content. Conversely, increasing levels of their mRNA can lead to increased seed oil accumulation. Particularly convincing are studies demonstrating that these genes, normally expressed predominantly in seeds, can induce the deposition of seed oil in vegetative tissues when ectopically activated in seedlings. Overexpression of the master regulators is also often associated with negative side effects. For example, seed-specific overexpression of maize LEC1 (ZmLEC1) resulted in an average 35% increase in seed oil but reduced germination and vegetative growth in the field. Conversely, modest (2- to 3-fold) seed-specific increases in mRNA levels of Brassica napus LEC1 (BnLEC1) or its closest relative, LEC1-LIKE (BnL1L), increased seed oil content by up to 20% (relative to untransformed controls) in replicated field trials without negative pleiotropic effects on growth, seed size, protein content, or nutritional value.
ABI3, LEC2 and FUS3 are closely related members of the plant-specific B3-domain family of transcription factors, constituting a specific subgroup sometimes referred to as the AFL clade. The B3-domain is so-named because it represents the third, and C-terminus proximal, basic domain of ABI3. This family of transcription factors has been shown to bind in vitro to the RY repeat/Sph element, which is enriched in promoters of seed-specific genes and possesses the minimal consensus sequence “CATGCA”. LEC1 belongs to a different family of transcription factors, constituting the HAP3 subunit of the trimeric CCAAT-box binding factor (CBF). Interestingly, LEC1 and L1L appear to be unique among Arabidopsis HAP3 subunits in their ability to regulate gene expression in a CBF-independent fashion. It is unclear which mechanism is relevant to LEC1’s role in seed oil synthesis.
The effects of LEC1 and FUS3 on seed oil content can be attributed, at least in part, to their positive regulation of several genes involved in the metabolism of carbohydrates and lipids. Of note, more than 50% of genes in the plastidial fatty acid synthesis pathway are targets of these two master regulators and levels of their mRNA increase within a few hours following chemical activation of inducible LEC1 or FUS3 transgenes. Included in this list are genes encoding several subunits of acetyl-CoA carboxylase, involved in the production of malonyl-CoA and the first committed step in fatty acid synthesis; acyl carrier protein (ACP), a plastidial cofactor in the Fatty Acid Synthase complex to which growing fatty acid chains are bound; and stearoyl-acyl carrier protein desaturase that catalyzes oleoyl-ACP formation. Other differentially regulated genes encode proteins involved in fatty acid modification (e.g. fatty acid desaturase 2 and fatty acid desaturase 3 that possess ω6 and ω3 desaturase activity, respectively; fatty acid elongases that increase the chain lengths of acyl-CoAs beyond 18 carbons), triacylglycerol synthesis (e.g. diacylglycerol acyltransferase that catalyzes the last step of triacylglycerol assembly), or oil packaging (e.g. oleosins which coat oil bodies). The master regulators also positively control the expression of genes implicated in sucrose synthesis and transport, and glycolysis, including several sucrose synthases and multiple subunits of plastidial pyruvate kinase and plastidial pyruvate dehydrogenase. These last two enzymes catalyze, respectively, the irreversible synthesis of pyruvate and its decarboxylation to acetyl-CoA for fatty acid synthesis. Increased expression of these carbohydrate-metabolism genes likely leads to increased carbon flux into fatty acid synthesis, a prerequisite to higher seed oil content.
Promoters of fatty acid synthesis genes are enriched for the consensus LEC1 (CBF) binding element but not the RY/Sph motif, suggesting that these genes may be direct targets of LEC1, but arguing for an indirect mechanism in the case of FUS3, albeit one involving a limited number of steps. Lack of direct interaction between B3-domain regulators and fatty acid synthesis genes is consistent with the identification of very few of these genes in large-scale transcript profiling analyses using inducible, ectopically expressed LEC2 genes. Given that the LEC2 studies focused on very short induction times compared to those tested with LEC1 and FUS3, only direct targets are likely to have been identified; it is nevertheless probable that LEC2 also participates indirectly to activate fatty acid synthesis genes. Conversely, the RY/Sph element is enriched in promoters of oleosin genes and several lines of evidence support direct regulation of these genes by the master regulators, in particular LEC2. First, transcripts of oleosins rapidly accumulate in seedlings following chemical induction of FUS3, LEC2 or LEC1; second, LEC2-induced oleosin expression does not require de novo protein synthesis, as it occurs in the presence of cycloheximide; and third, LEC2 specifically binds in vitro to RY/Sph elements located in oleosin promoters.
As previously mentioned, the AFL clade B-domain factors and LEC1 regulate numerous aspects of seed development, and ectopic expression of LEC1 or LEC2 induces the formation of somatic embryos. However, results summarized above are consistent with the more direct involvement of the master regulators in regulating seed oil accumulation through the expression of lipid metabolism genes. Indeed, increased oil synthesis does not appear to be an indirect consequence attributed to the natural progression of somatic embryos through the maturation phase.
Interactions between the master regulators are complex and do not adhere to a strict hierarchical pattern. When ectopically expressed in seedlings, LEC1 and LEC2 positively regulate the expression of FUS3 and ABI3. LEC2 also up-regulates LEC1 expression, but the opposite is not true. LEC1-mediated fatty acid synthesis gene expression and seed oil accumulation is dependent on FUS3 and, to a lesser extent, ABI3; however, the requirement for other master regulators upon inducible ectopic expression has not been reported for LEC2. LEC2 expression is also positively regulated by at least two other groups of transcription factors; the MADS-box protein AGAMOUS-LIKE15 (AGL15) and BASIC PENTACYSTEINE (BPC) proteins. Interestingly, AGL15 is also a direct target of LEC2.
Ironing Out WRINKLED Control of Glycolysis and Fatty Acid Synthesis
One level below the master regulators in the transcriptional cascades that control seed oil deposition is WRINKLED1 (WRI1), a member of the large, plant-specific APETALA2 (AP2) family of transcription factors. The expression of WRI1 is up-regulated by LEC1, LEC2 and FUS3, and the gene is a direct target of LEC2, and possibly of FUS3. The WRI1 gene is so-named because the Arabidopsis mutant produces wrinkled seeds with 20% of wild-type oil content and increased starch levels. Seed oil in wri1 mutants is also altered in fatty acid composition, with higher proportions of linolenic acid (18:3cisΔ9,12,15) and to a lesser extent, erucic acid (22:1cisΔ13) at the expense of oleic (18:1cisΔ9), linoleic (18:2cisΔ9,12) and eicosenoic (20:1cisΔ11) acids. Overexpression of WRI1 elevates relative oil levels in seeds by up to 20% and constitutive WRI1 expression can induce ectopic seed oil synthesis in vegetative tissues when seedlings are grown on sucrose-containing media. Increased seed oil in WRI1 transgenics does not appear to be generally correlated with altered fatty acid composition, although complementation of the Arabidopsis wri1 mutant by either of two maize homologs displayed increased 18:3 with reduced 18:1 and 18:2. In B. napus, expression levels of WRI1 genes were correlated with seed oil content in different genotypes, and a WRI1 polymorphism was shown to be significantly associated with oil content in a segregating population. In maize, the two known WRI1 homologues display unique patterns of gene expression and it has been proposed that one is dedicated to plastidial fatty acid synthesis for seed oil production while the other may play a general role in triacylglycerol synthesis.
With one exception, only minor negative side effects have been associated with WRI1 overexpression. In a detailed study that included replicated, multi-site field testing of elite hybrids, transgenic expression of a maize WRI1 homolog from an embryo-specific promoter increased oil content in grains by 10-22%, with a concomitant reduction in starch levels (60%), but no change in protein content. These transgenic plants did not display any notable effects on plant development, including germination, flowering time, or grain weight. An independent study using greenhouse-grown maize reported very similar findings. Expression of two Brassica WRI1 homologues in Arabidopsis resulted in increased seed oil with no change in protein levels, and substantial increases in seed weight (up to 40%), which was attributed to the production of bigger embryos consisting of larger cells. The sizes of siliques and the number of seeds per silique were not affected. In contrast, overexpression in Arabidopsis of the endogenous WRI1 mRNA increased oil content but did not affect the number of seeds produced per plant. The possible effects on seed size were not reported. In a different study, constitutive overexpression of the Arabidopsis WRI1 (again in Arabidopsis) yielded numerous pleiotropic effects, ranging from stunted growth and reduced apical dominance, to poor seed set, reduced seed size and reduced seed oil content. Expression of WRI1 using a seed-specific promoter alleviated these effects, but did not lead to increases in seed oil content, despite the presence of much higher levels of WRI1 mRNA. The reason for these distinct differences in results between studies remains to be resolved, but may be attributed to choice of promoter, genotype, and growth conditions. Constitutive expression of WRI1 has also been reported to have a negative impact on germination in the presence of sucrose.
WRI1’s primary function in seed oil deposition appears to be the positive regulation of genes encoding enzymes involved in late glycolysis and fatty acid biosynthesis. Expression of these genes is reduced in seeds of wri1 mutants and increased in overexpressing lines. Genes differentially regulated in both Arabidopsis and maize, thus representing conserved WRI1 targets, include those encoding several subunits of each plastidial pyruvate kinase and plastidial pyruvate dehydrogenase, ACP, ketoacyl-ACP synthase III, and acyl-ACP thio-esterase (FAT A). Genes encoding several subunits of plastidial acetyl-CoA carboxylase were found to be differentially expressed only in Arabidopsis, while those encoding mitochondrial ketopantoate hydroxymethyltransferase and carbonic anhydrase, both involved in the synthesis of acetyl-CoA, were up-regulated only in maize. It remains possible that these genes are conserved targets of WRI1 but failed to be identified in both species due to experimental design. Consistent with the altered patterns of gene expression, the activities of several glycolytic enzymes are compromised in developing wri1 seeds and flux through plastidial pyruvate kinase, which provides most of the carbon for de novo fatty acid biosynthesis, is dramatically reduced. Transcript levels for genes involved in later steps of seed oil metabolism, such as triacylglycerol assembly and packaging, are reduced in wri1 mutants but appear to be unaffected by WRI overexpression in either Arabidopsis or maize. In Arabidopsis, WRI1 also up-regulates the expression of the GLB1 gene which encodes a protein (PII) capable of inhibiting plastidial acyl-CoA carboxylase, which is encoded by genes that are themselves regulated by WRI1. Through PII, WRI may integrate carbon and nitrogen metabolism, and energy levels in seeds.
Many WRI1-regulated genes are also under the control of the master regulators. Seed oil accumulation in seedlings ectopically expressing LEC1 or LEC2 is dependent on WRI1, indicating that the seed oil-related function of these master regulators is largely mediated through WRI1. However, overexpression of WRI1 does not fully restore the seed oil content of the lec2 mutant, suggesting the requirement of other factors in regulating LEC2-mediated seed oil synthesis. Given that induction of LEC1 in seedlings did not activate LEC2 expression, LEC1 and LEC2 may control two parallel pathways towards WRI1 activation.
The consensus DNA-binding motif for WRI1, the AW box [(CnTnG](n)7[CG)], is enriched in promoters of fatty acid biosynthesis genes. WRI1 has been shown to bind directly to the AW box of several fatty acid synthesis genes in vitro, and mutation of the AW box abolishes WRI-mediated transcriptional activation in transient protoplast assays. WRI1 was also shown to transactivate the FATTY ACID ELONGASE1 gene, although the requirement for the AW box was not elucidated. WRI1 did not activate either TAG1, encoding diacylglycerol acyltransferase, or OLEOSIN. Together, these data indicate that, in addition to WRI1, other key transcription factors are required for the activation of genes involved in triacylglycerol and oil body assembly.
WRI1 has also been implicated in the control of plastidial fatty acid synthesis in the mesocarp of oil palm, which, although not a seed, accumulates very high levels of triacylglycerol (up to 90%). Transcript levels of a putative WRI1 gene from oil palm are substantially higher (57-fold) in the mesocarp than in its relative date palm, which does not accumulate triacylglycerol. Levels of plastidial fatty acid gene transcripts were also elevated in oil palm, on average 13-fold, and their temporal expression profile mirrored that of WRI1. Transcripts of genes involved in triacylglycerol assembly were similar between oil and date palm mesocarp, suggesting that factors other than WRI1 are involved in the regulation of these pathways. Interestingly, expressed sequence tags (ESTs) of the seed maturation master regulators were not detected in either oil or date palm mesocarp, indicating a possible unique mode of WRI1 activation in these tissues.
DOF’s Oily Finger
Searches for putative cis-element binding motifs enriched in promoters of fatty acid synthesis genes suggest the involvement of DOF, GATA, and MYB classes of transcription factors, while searches of oleosin promoters identified cognate binding sites for basic leucin zipper (bZIP) factors. DOF (DNA binding with one finger) and GATA transcription factors both contain zinc finger motifs. Analysis of several DOF factors from soybean (Glycine max) identified two (GmDof4 and GmDof11) capable of enhancing seed oil content and increasing seed weight when constitutively expressed in Arabidopsis. Transgenic expression of GmDof4 decreased the proportion of 18:2 in seed oil, while GmDof11 did not alter fatty acid composition. Both of these transcription factors are capable of binding in vitro to cognate DOF cis-elements in the promoters of genes encoding the β-subunit of acetyl-CoA carboxylase and a long-chain acyl-CoA synthetase, respectively. These genes are differentially expressed in the respective transgenic lines, suggesting that they are direct targets of the DOF proteins. Levels of seed storage proteins may be negatively regulated by GmDof4 and GmDof11, as these bind to the promoter of a gene (CRA1) encoding a seed storage protein which is down-regulated in the transgenic seeds.
Slimy Exterior: the Indirect Role of GLABRA2
GLABRA2 (GL2), a member of the homeodomain-leucine zipper (HD-ZIP) family of transcription factors, was originally identified as a regulator of epidermal cell fate, specifically trichome development. Mutation of Arabidopsis GL2 or antisense suppression of BnGL2 in canola lead to increases in seed oil, implicating this transcription factor as a negative regulator. To this end, GL2 activity is required in the seed coat but not in the embryo, and does not require the putative target gene PHOSPHOLIPASE D ZETA 1. GL2 also regulates the expression of MUCILAGE MODIFIED 4 (MUM4), which encodes a rhamnose synthase and is required for seed mucilage formation, as is GL2 itself. Loss of MUM4 and other genes required for mucilage formation also result in increased seed oil content, suggesting that the phenomenon is related to greater carbon availability in the absence of mucilage. However, a mutant at the TTG2 locus, which encodes a WRKY class of transcription factor and is defective in mucilage formation, is not compromised in MUM4 expression and does not have elevated seed oil. It was speculated that in the ttg2 mutant, carbon may already be committed to rhamnose and no longer readily diverted to fatty acid synthesis.
The AP2 transcription factor also has been shown to act maternally, as a negative regulator of seed size. Arabidopsis ap2 mutants produce larger seeds with concomitant increases in seed oil. The mutants display a longer maturation phase and higher concentrations of hexoses in maturing seeds than the wild-type. One possibility is that AP2 modulates seed oil synthesis indirectly through the sugar metabolism in the seed coat.
Negative Vibe: Repressing Seed Oil Biosynthesis in Vegetative Tissues
Given that several of the transcription factors discussed above have strong positive roles in promoting seed-specific programs, mechanisms have evolved to ensure that their expression is suppressed during vegetative growth. Genes contributing to this suppression have been identified as loss-of-function mutants that ectopically express the master regulators outside of the seed. These suppressors all appear to be involved in determining the chromatin conformation of target genes. They have generated interest as a possible means to metabolically engineer storage lipids in vegetative tissues such as leaves, tubers and roots to increase their energy index.
The first suppressor to be characterized was PICKLE (PKL), which encodes a putative chromatin remodeling factor belonging to the chromodomain/helicase/DNA-binding (CHD3) family. Levels of LEC1, LEC2 and FUS3 mRNA are close to 1000-fold higher in roots of pkl mutants than wild-type plants, leading to the ectopic expression of embryogenic genes, the synthesis of storage lipids and swollen green primary root tips. CHD3 proteins are known to act as transcriptional repressors, often as part of complexes possessing histone deacetylase activity. However, chromatin of pkl mutants shows no differences in histone acetylation but is depleted in trimethylated histone H3 lysine 27 (H3K27m3), a modification widely associated with gene silencing. In germinating wild-type seeds, PKL is physically associated with promoters enriched in H3K27m3, including LEC1, LEC2, and FUS3, suggesting that it acts to directly promote this repressive chromatin mark on the genes encoding master regulators of seed maturation at the transition from seed to vegetative growth. PKL function appears to be transient, and is no longer required two weeks following germination in Arabidopsis.
The H3K27m3 mark is deposited by Polycomb Repressor Complex 2 (PRC2) and there are conflicting reports on the role these proteins play in mediating PKL function. It is clear, however, that PCR2 contributes to repressing the master seed regulators in vegetative plant tissues, as mutation of several of its components result in the derepression of master regulator genes and the production of seed oil in seedlings. A cis-acting element called RLE has been characterized in the promoter of LEC2 that is required for deposition of H3K27m3, and may be analogous to Drosophila PRE, which acts to recruit PRC2 complexes.
A clade of B3-domain transcription factors, separate but related to the AFL clade discussed above, also acts redundantly to suppress seed maturation programs in vegetative tissues. Double mutants in two of these factors, HIGH-LEVEL EXPRESSION OF SUCROSE INDUCIBLE GENE2 (HSI2), also known as VIVIPAROUS AND ABI3-LIKE1 (VAL1), and HSI2-LIKE (HSIL or VAL2), produce seedlings expressing AFL clade B3-domain genes and large numbers of embryo-specific genes containing Sph/RY elements in the promoters, accumulate seed storage compounds and exhibit ectopic embryo formation. In addition to the B3 domain, HSI2/VAL proteins contain domains associated with chromatin remodeling factors and transcriptional repressors, and HSI2 has been shown to be a potent transcriptional repressor in vitro.
ARABIDOPSIS SIX-B-INTERACTING PROTEIN 1-LIKE 1 (ASIL1) represents another suppressor of B3-domain master regulators that accumulates storage lipids in seedling tissues upon mutation. ASIL1 also contributes to the expression of AFL clade B3-domain genes and an oleosin gene during seed maturation. Although it displays a number of developmental abnormalities, the asil1 mutant does not produce pkl roots or ectopic embryos reminiscent of the pkl or val mutants.
Closing Comments
WRI1 and the master regulators of seed maturation are presently the best understood transcription factors that contribute in seed oil deposition. There are certainly additional transcription factors involved, acting either directly on lipid biosynthetic genes or indirectly to influence seed size, development or the allocation of resources. Of particular relevance are the further characterization of WRI1 function and the identification of other transcription factors that act specifically on different steps of lipid biosynthesis. Rapid developments in the field of genomics, for example the emergence of next-generation sequencing technologies, should facilitate the discovery of transcription factors in crop species and their associated transcriptional cascades.
Some Key References
- Baud, S., Bourrellier, A.B.F., Azzopardi, M., Berger, A., Dechorgnat, J., Daniel-Vedele, F., Lepiniec, L., Miquel, M., Rochat, C., Hodges, M. and Ferrario-Méry, S. PII is induced by WRINKLED1 and fine-tunes fatty acid composition in seeds of Arabidopsis thaliana. Plant J., 64, 291–303 (2010) (DOI: 10.1111/j.1365-313X.2010.04332.x).
- Baud, S. and Lepiniec, L. Physiological and developmental regulation of seed oil production. Prog. Lipid Res., 49, 235–249 (2010) (DOI: 10.1016/j.plipres.2010.01.001).
- Baud, S., Wuilleme, S., To, A., Rochat, C. and Lepiniec, L. Role of WRINKLED1 in the transcriptional regulation of glycolytic and fatty acid biosynthetic genes in Arabidopsis. Plant J., 60, 933–947 (2009) (DOI: 10.1111/j.1365-313X.2009.04011.x).
- Berger, N., Dubreucq, B., Roudier, F., Bubos, C. and Lepiniec, L. Transcriptional regulation of Arabidopsis LEAFY COTYLEDON2 involves RLE, a cis-element that regulates trimethylation of histone H3 at lysine-27. Plant Cell, 23, 4065-4078 (2011) (DOI: 10.1105/tpc.111.087866).
- Bourgis, F., Kilaru, A., Cao, X., Ngando-Ebongue, G.F., Drira, N., Ohlrogge, J.B. and Arondel, V. Comparative transcriptome and metabolite analysis of oil palm and date palm mesocarp that differ dramatically in carbon partitioning. Proc. Natl. Acad. Sci. U.S.A., 108, 12527-12532 (2011) (DOI: 10.1073/pnas.1106502108).
- Braybrook, S.A. and Harada, J.J. LECs go crazy in embryo development. Trends Plant Sci., 13, 624-630 (2008) (DOI: 10.1016/j.tplants.2008.09.008).
- Gao, M.J., Lydiate, D.J., Li, X., Lui, H., Gjetvaj, B., Hegedus, D.D. and Rozwadowski, K.Repression of seed maturation genes by a trihelix transcriptional repressor in Arabidopsis seedlings. Plant Cell, 21, 54-71 (2009) (DOI: 10.1105/tpc.108.061309).
- Liu, J., Hua, W., Zhan, G., Wei, F., Wang, X., Liu, G. and Wang, H. Increasing seed mass and oil content in transgenic Arabidopsis by the overexpression of a wri1-like gene from Brassica napus. Plant Physiol. Biochem., 49, 9-15 (2009) (DOI: 10.1016/j.plaphy.2009.09.007).
- Maeo, K., Tokuda, T., Ayame, A., Mitsui, N., Kawai, T., Tsukagoshi, H., Ishiguro, S. and Nakamura, K. An AP2-type transcription factor, WRINKLED1, of Arabidopsis thaliana binds to the AW-box sequence conserved among proximal upstream regions of genes involved in fatty acid synthesis. Plant J., 60, 476–487 (2009) (DOI: 10.1111/j.1365-313X.2009.03967.x).
- Mu, J., Tan, H., Zheng, Q., Fu, F., Liang, Y., Zhang, J., Yang, X., Wang, T., Chong, K., Wang, X.J. and Zuo, J.LEAFY COTYLEDON1 is a key regulator of fatty acid biosynthesis in Arabidopsis. Plant Physiol., 148, 1042-1054 (2008) (DOI: 10.1104/pp.108.126342).
- Ohto, M., Floyd, S.K., Fischer, R.L., Goldberg, R.B. and Harada, J.J. Effects of APETALA2 on embryo, endosperm, and seed coat development determine seed size in Arabidopsis. Sex. Plant Repro., 22, 277-289 (2009) (DOI: 10.1007/s00497-009-0116-1).
- Peng, F.Y. and Weselake, R.J. Gene coexpression clusters and putative regulatory elements underlying seed storage reserve accumulation in Arabidopsis. BMC Genomics, 12, 286 (2011) (DOI: 10.1186/1471-2164-12-286).
- Pouvreau, B., Baud, S., Vernoud, V., Morin, V., Py, C., Gendrot, G., Pichon, J.P., Rouster, J., Paul, W. and Rogowsky, P.M. Duplicate maize Wrinkled1 transcription factors activate target genes involved in seed oil biosynthesis. Plant Physiol., 156, 674-686 (2011) (DOI: 10.1104/pp.111.173641).
- Shen, B., Allen, W.B., Zheng, P.Z., Li, C.J., Glassman, K., Ranch, J., Nubel, D. and Tarczynski, M.C. Expression of ZmLEC1 and ZmWRI1 increases seed oil production in maize. Plant Physiol., 153, 980–987 (2010) (DOI: 10.1104/pp.110.157537).
- Shi, L., Katavic, V., Yu, Y., Kunst, L. and Haughn, G. Arabidopsis glabra2 mutant seeds deficient in mucilage biosynthesis produce more oil. Plant J., 69, 37-46 (2012) (DOI: 10.1111/j.1365-313X.2011.04768.x).
- Suzuki, M. and McCarty, D.R. Functional symmetry of the B3 network controlling seed development. Curr. Opin. Plant Biol., 11, 548-553 (2008) (DOI: 10.1016/j.pbi.2008.06.015).
- Tan, H., Yang, X., Zhang, F., Zheng, X., Qu, C., Mu, J., Fu, F., Li, J., Guan, R., Zhang, H., Wang, G. and Zuo, J. Enhanced seed oil production in canola by conditional expression of Brassica napus LEAFY COTYLEDON1 and LEC1-LIKE in developing seeds. Plant Physiol., 156, 1577-1588 (2011) (DOI: 10.1104/pp.111.175000).
- Wang, H.W., Zhang, B., Hao, Y.J., Huang, J., Tian, A.G., Liao, Y., Zhang, J.S. and Chen, S.Y. The soybean Dof-type transcription factor genes, GmDof4 and GmDof11, enhance lipid content in the seeds of transgenic Arabidopsis plants. Plant J., 53, 716–729 (2007) (DOI: 10.1111/j.1365-313X.2007.03268.x).
- Yamamoto, A., Kagaya, Y., Usui, H., Hobo, T., Takeda, S. and Hattori, T. Diverse roles and mechanisms of gene regulation by the Arabidopsis seed maturation master regulator FUS3 revealed by microarray analysis. Plant Cell Physiol., 51, 2031-2046 (2010) (DOI: 10.1093/pcp/pcq162).
- Zhang, H., Bishop, B., Ringenberg, W., Muir, W.M. and Ogas, J. The CHD3 remodeler PICKLE associates with genes enriched for trimethylation of histone H3 lysine 27. Plant Physiol., 159, 418-432 (2012) (DOI: 10.1104/pp.112.194878).
In This Section
- Plant Fatty Acid Synthesis
- Production of Unusual Fatty Acids in Plants
- Arabidopsis Acyl-Coenzyme A-Binding Proteins
- Long Chain acyl-coA Synthetases and Other Acyl Activating Enzymes
- Plant Triacylglycerol Synthesis
- Triacylglycerol Biosynthesis in Eukaryotic Microalgae
- Subcellular Oil Droplets and Oleosins in Plants
- Triacylglycerol Mobilisation in Plants
- Role of Transcription Factors in Storage Lipid Accumulation in Plants
- Biosynthesis of Plant Lipid Polyesters
- Rubber Biosynthesis in Plants
- Carotenoid Biosynthesis and Regulation in Plants
- The Oxylipin Biosynthetic Pathways in Plants
- N-Acylphosphatidylethanolamines (NAPEs), N-acylethanolamines (NAEs) and Other Acylamides: Metabolism, Occurrence and Functions in Plants
- Phosphoinositide Signaling in Plants
- Plant Lipidomics
- 50 years of Galactolipid Research: The Beginnings
- Transport and function of lipids in the plant phloem