Mammalian Diacylglycerol Acyltransferases (DGAT)
The Author: Scot J. Stone, Department of Biochemistry, Room 14.1 Health Sciences Building, University of Saskatchewan, 107 Wiggins Road, Saskatoon, SK, Canada, S7N 5E5. DOI: 10.21748/lipidlibrary.39186
Introduction
Triacylglycerols (TGs) are esters of fatty acids and glycerol and are the major form of stored energy in eukaryotic organisms. TGs are a normal and necessary component of mammalian energy homeostasis. However, an imbalance between energy intake and expenditure can lead to the excessive accumulation of TG in tissues with pathological consequences [1,2]. Accumulation of TGs in adipose tissue can lead to obesity. In nonadipose tissues, such as skeletal muscle, liver, heart, and pancreatic β-cells, excessive levels of TGs are associated with insulin resistance and can lead to type 2 diabetes mellitus. Increased TGs in the blood are a risk factor for atherosclerosis. Almost all mammalian tissues are capable of synthesizing TGs. The tissues with the highest TG content generally include adipose tissue, liver, mammary gland and the small intestine, all of which have central roles in energy metabolism.
Triacylglycerol Biosynthesis
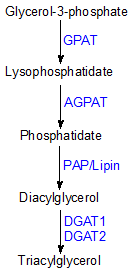
TG synthesis occurs via the multistep glycerol-3-phosphate or Kennedy pathway (Figure 1) [3,4].
Figure 1. Triacylglycerol biosynthesis via the glycerol-3-phosphate (Kennedy) pathway
(GPAT, glycerol-3-phosphate acyltransferase; AGPAT, 1-acylglycerol-3-phosphate acyltransferase; PAP/lipin, phosphatidate phosphatase; DGAT, diacylglycerol acyltransferase)
Glycerol 3-phosphate, derived primarily from glycolysis or gluconeogenesis, is esterified at the sn-1 carbon position by glycerol 3-phosphate acyltransferases producing lysophosphatidate (1-acylglycerol 3-phosphate). 1-Acylglycerol-3-phosphate acyltransferase catalyzes the esterification of the sn-2 carbon of lysophosphatidate forming phosphatidate. The phosphatidate phosphatases (lipins) then dephosphorylate phosphatidate producing 1,2-diacylglycerol, which can then be used to synthesize either phospholipids or TG. Diacylglycerol can also be generated via the monoacylglycerol acyltransferase (MGAT) pathway [5]. In this pathway, 1,2-diacylglycerol is formed by the direct acylation of 2-monoacylglycerol at the sn-1 position. This pathway provides 80-85% of the TG synthesized by enterocytes during intestinal fat absorption. The last step of the TG biosynthetic pathway is catalyzed by acyl coenzyme-A:1,2-diacylglycerol acyltransferase (DGAT) at the endoplasmic reticulum. DGAT catalyzes the synthesis of TGs by forming an ester bond between a fatty acid, donated from a long-chain fatty acyl CoA, and the free hydroxyl group at the sn-3 position of 1,2-diacylglycerol (Figure 2).
Figure 2. DGAT catalyzes the acyl-CoA-dependent synthesis of triacylglycerol.
Roles of DGAT in Triacylglycerol Metabolism
The role of DGAT in TG metabolism should be considered in the context of individual tissues:
Small intestine - In the small intestine, DGAT is required for the absorption of dietary TGs. Dietary TGs are nonpolar molecules and are not able to be transported from the intestinal lumen to enterocytes intact. Instead, TGs in the small intestine are emulsified and digested by lipases producing 2-monoacylglycerol and unesterified fatty acids, which cross from the lumen of the gut into enterocytes. In the enterocyte, TGs are resynthesized mainly by monoacylglycerol acyltransferase and DGAT. TGs are incorporated into chylomicrons in order to deliver dietary lipids through the lymphatic system to the circulation where the fatty acids are taken up by muscle, liver, adipose tissue, etc.
Liver – In the liver, DGAT has a role in synthesizing TGs from either fatty acids synthesized de novo or from fatty acids taken up from the circulation. These TGs are incorporated into very-low-density lipoproteins for delivery to extrahepatic tissues where they are stored (adipose tissue) or oxidized (skeletal and cardiac muscle).
Mammary gland – TGs, a major component of milk, are stored in adipocytes in the lactating mammary gland and provide an essential source of energy to newborns. Fatty acids released by the hydrolysis of TGs stored in adipose tissue are reesterified to TGs by DGAT in the mammary gland.
Adipose tissue – Adipose tissue has the highest content of TGs in mammals and is the main tissue for storage of TG. TGs are delivered to adipose tissue through the circulation by chylomicrons and very-low-density lipoproteins (VLDL). Lipoprotein lipase present in the blood capillaries in adipose tissue hydrolyzes TGs contained within these lipoproteins. The unesterified fatty acids are taken up by adipocytes, re-esterified to TGs mainly by the Kennedy pathway involving DGAT and stored in cytosolic lipid droplets. When required, TGs in adipose tissue are hydrolyzed to fatty acids and glycerol, which are released into the circulation. Fatty acids are then transported in an albumin-bound form to tissues such as muscle and liver where they are oxidized to promote the synthesis of ATP.
Multiple DGAT Enzymes
DGAT activity was first described by Weiss and Kennedy in the 1950s [3,4]. Most attempts to purify this enzyme to homogeneity so that it could be studied in isolation were largely unsuccessful. Upon the advent of the molecular biological era, two DGAT enzymes were identified, DGAT1 and DGAT2 [6,7]. These enzymes have no sequence homology whatsoever. Further discussion will focus on the current knowledge of DGAT1 and DGAT2 in mammalian systems. However, orthologues of both enzymes exist in a wide variety of species, including plants, fungi, and invertebrates (see our web page on triacylglycerol biosynthesis in plants, for example).
DGAT1
DGAT1 belongs to a large family of membrane-bound O-acyltransferases (MBOAT) and was initially discovered by its homology to acyl CoA:cholesterol acyltransferase (ACAT)-1 and -2, which catalyze cholesterol ester biosynthesis [6]. DGAT1 is ubiquitously expressed in human tissues, but is most abundant in the small intestine, adipose tissue, mammary gland, liver and muscle.
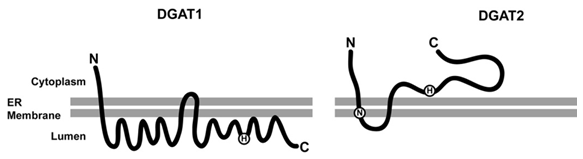
DGAT1 is a 55 kDa integral membrane protein that is localized to the endoplasmic reticulum. Originally predicted to have 6–12 possible transmembrane domains, recent evidence suggests that DGAT1 has only three, with its N-terminus exposed to the cytosol and its C-terminus present in the lumen of the endoplasmic reticulum (Fig. 3) [8]. The C-terminal region of DGAT1 in the ER lumen contains a highly conserved histidine (H426 of murine DGAT1) that is essential for DGAT1 function and is likely part of the active site. The luminal orientation of the active site suggests a role for DGAT1 in very-low-density lipoprotein (VLDL) production in the liver and chylomicron production in the small intestine. TG synthesized in this region of the endoplasmic reticulum could be accessible to the cellular machinery responsible for the synthesis and secretion of lipoproteins. Overexpression of DGAT1 in McArdle RH7777 rat hepatoma cells stimulated TG synthesis and VLDL secretion [9]. A study in mice showed that DGAT1 overexpressed in liver via adenovirus infection increased hepatic TG levels and VLDL secretion [10]. Inhibition of DGAT1 using a small molecule inhibitor significantly reduced plasma TG levels in hyperlipidemic rat and hamster models [11].
Figure 3. Topological orientation of DGAT1 and DGAT2.
ⒽActive site histidine,ⓃNeutral lipid binding domain.
DGAT1 appears to exist as a dimer and tetramer with the former form likely being the minimal catalytic unit [8,12]. The N-terminal region of DGAT1 appears to be involved in tetramer formation and the regulation of DGAT activity. Removal of the N-terminus of DGAT1 prevented tetramer formation and corresponded with a severalfold increase in in vitro enzyme activity.
In addition to catalyzing the synthesis of TGs, DGAT1 is also able to synthesize diacylglycerol, retinol and wax esters, in vitro, via its acyl-CoA:monoacylglycerol acyltransferase (MGAT), wax synthase and acyl-CoA:retinol acyltransferase (ARAT) activities [13]. The relevance of these additional acyltransferase activities in vivo is not clear. The most information available is in regards to the acyl-CoA:retinol acyltransferase activity of DGAT1 in the skin where mice lacking DGAT1 were found to have reduced acyl-CoA:retinol acyltransferase activity [14]. In the absence of DGAT1, the inability to synthesize retinol esters led to retinoid toxicity and hair loss. It appears that in addition to its role in TG metabolism, DGAT1 also functions as the major acyl-CoA:retinol acyltransferase enzyme in the skin of mice and has a significant role maintaining retinoid homeostasis.
DGAT2
DGAT2 was initially purified from the lipid body fraction from the fungus Mortierella ramanniana [15]. Several homologous mammalian sequences were subsequently identified and collectively grouped into the DGAT2 gene family [7]. Expression of one of these sequences encoding a 44 kDa protein in insect cells confirmed the identity of the second DGAT enzyme (DGAT2). DGAT2 is expressed in most mammalian tissues, with the highest expression levels in lipogenic tissues such as mammary gland, liver, small intestine and adipose tissue. Unlike DGAT1, DGAT2 does not possess any other acyltransferase activities.
DGAT2 is an integral membrane protein found mostly in the endoplasmic reticulum. When TG synthesis in cells is stimulated by supplementing growth medium with fatty acids, DGAT2 becomes concentrated around the surface of cytosolic lipid droplets [16,17]. Immunogold electron microscopy and subcellular fractionation showed that DGAT2 was present on lipid droplets. However, this accounted for only a small fraction of total cellular DGAT2 protein. A more likely explanation is that DGAT2 is present in ER membranes in close proximity to lipid droplets.
DGAT2 has two transmembrane domains with both the N- and C-termini facing the cytosol (Fig. 3) [18]. Two highly conserved sequences present in DGAT2 are required for enzymatic function. “His-Pro-His-Gly” is present in all DGAT2 family members and is likely part of the active site. Its cytosolic orientation may facilitate the transfer of newly synthesized TGs from the endoplasmic reticulum membrane to its site of storage - cytosolic lipid droplets. The second domain is a neutral lipid binding domain (FLXLXXXn; n is a nonpolar amino acid) that is present in other proteins involved in lipid metabolism, such as cholesterol ester transfer protein, hormone-sensitive lipase, lecithin:cholesterol acyltransferase, cholesterol 7α-hydroxylase, cholesterol esterase, and TG hydrolase. Neither of these domains is present in DGAT1.
DGAT2 has a central role in intracellular TG accumulation. When overexpressed in McArdle RH7777 rat hepatoma and COS-7 cells, DGAT2 more potently stimulated TG synthesis than DGAT1, yielding a much larger increase in intracellular TG, which accumulated in large cytosolic lipid droplets [16,17,19].
Regulation of DGAT Enzymes
Adipogenesis - Both DGAT1 and DGAT2 gene expression increase during adipogenesis which is accompanied by an increase in total DGAT activity and TG synthesis [6,7]. Although direct evidence is lacking, it appears that DGAT1 and DGAT2 are likely regulated by C/EBPα or PPARγ, key transcriptional regulators of adipogenesis.
Hormonal Regulation
Available evidence suggests that DGAT1 and DGAT2 expression are under hormonal control. Addition of glucose or insulin to the growth medium of 3T3-L1 adipocytes increased in vitro DGAT activity [20]. Both DGAT1 and DGAT2 expression was stimulated by glucose, while only DGAT2 expression was stimulated by insulin. Additionally, DGAT2 mRNA levels in adipose tissue and liver are decreased by fasting and increased following refeeding. This indicates that TG synthesis by DGAT2 can be modulated by energy status.
In mouse models of obesity, DGAT1 and DGAT2 gene expression appears to be reciprocally regulated [13]. Increased DGAT2 expression in adipose tissue is likely responsible for the increased deposition of TG leading to obesity. In contrast, DGAT1 expression is decreased, although the significance of this is not clear. Leptin, a peptide hormone secreted by adipose tissue that regulates food intake and energy expenditure, also appears to have a role in regulating DGAT2 expression. Mice lacking a functional leptin pathway have increased DGAT2 expression promoting increased TG synthesis and adiposity [21].
Post-Transcriptional Regulation
Studies performed using rat liver microsomes demonstrated that in vitro DGAT activity could be regulated by a phosphorylation-dephosphorylation mechanism [22]. Microsomal membranes isolated in the presence of fluoride, a phosphatase inhibitor, had lower DGAT activity compared to microsomes isolated without fluoride. DGAT activity of rat hepatocytes and adipose tissue could be inactivated by a cytosolic factor in the presence of ATP and Mg2+, implying that a kinase in the cytosol can inhibit DGAT activity. Studies with kinase inhibitors suggested that this cytosolic factor is a tyrosine kinase. DGAT1, but not DGAT2, possesses a putative tyrosine phosphorylation site. However, mutation of this tyrosine in human DGAT1 has no effect on in vitro DGAT activity or TG synthesis [23]. Thus, it appears unlikely that tyrosine phosphorylation at this site regulates DGAT1 activity.
Hamster fibroblasts incubated with cell-permeable cAMP analogues increase TG synthesis, which is attributed to increased DGAT activity, suggesting regulation by cAMP-dependent protein kinase A (PKA) [13]. In a separate study, cAMP inhibited DGAT activity in rat hepatocytes [13]. It is possible that DGAT activity responds differently to cAMP in a tissue-specific manner. DGAT1, and not DGAT2, has the consensus sequence for two PKA phosphorylation sites (T15 and S244), suggesting that DGAT1 could be regulated by PKA.
Roles of DGAT1 and DGAT2 in Triacylglycerol Metabolism in vivo
Mouse models lacking or overexpressing DGAT1 and DGAT2 have been developed to investigate the roles of these two enzymes in lipid metabolism in vivo. Studies in DGAT1 and DGAT2 knockout mice have provided definitive evidence that these two enzymes do not serve redundant functions and have distinct roles in TG metabolism.
DGAT1-Deficient Mice
Mice lacking DGAT1 (Dgat1–/–) are viable with less adipose mass (~50% reduction) [13,24]. These mice are leaner than wild-type mice, and are resistant to diet-induced obesity, through a mechanism involving increased energy expenditure (physical activity and thermogenesis). TG levels in tissues such as the liver and skeletal muscle are moderately reduced in Dgat1–/– mice and plasma TG levels are normal. Dgat1–/– mice also have improved glucose metabolism as they have increased insulin sensitivity.
Although highly expressed in the small intestine, DGAT1 is not required for the quantitative absorption of dietary TG or chylomicron synthesis. However, the rate of TG absorption in the intestine is delayed in the absence of DGAT1. Other mechanisms (DGAT2 and/or diacylglycerol transacylase) likely compensate for the absence of DGAT1 in the intestine.
DGAT2-Deficient Mice
Unlike DGAT1, DGAT2 was found to be essential for life [13,19]. DGAT2-deficient (Dgat2–/–) mice die in the early postnatal period, with newborn pups surviving only 6–8 h after birth. The carcass TG content of Dgat2–/– mice is reduced by ~90% suggesting that insufficient energy stores were available to support survival after birth. An unexpected finding was that Dgat2–/– mice have severe skin abnormalities in which there is increased transepidermal water loss leading to rapid dehydration. The skin abnormalities observed were likely a result of essential fatty acid deficiency leading to decreased acylceramide levels in the skin. Acylceramide is synthesized from linoleic acid and is essential for maintaining the skin permeability barrier.
Concluding Remarks
The identification of genes encoding DGAT1 and DGAT2 has allowed significant progress to be made in understanding the fundamental process of TG synthesis. This knowledge should be of clinical importance with respect to the development of therapies for the treatment or prevention of diseases resulting from the excessive accumulation of TG in tissues or the circulation.
References
- Unger, R.H. Lipotoxic diseases. Annu. Rev. Med., 53, 319–336 (2002).
- Rosenbaum, M., Leibel, R. L. and Hirsch, J. Obesity. New Engl. J. Med., 337, 396–406 (1997).
- Kennedy, E.P. Metabolism of lipides. Annu. Rev. Biochem., 26, 119-148 (1957) (DOI: 10.1146/annurev.bi.26.070157.001003).
- Weiss, S.B. and Kennedy, E.P. The enzymatic synthesis of triglycerides. J. Am. Chem. Soc., 78, 3550 (1956).
- Senior, J.R. and Isselbacher, K.J. Direct esterification of monoglycerides with palmityl coenzyme A by intestinal epithelial subcellular fractions. J. Biol. Chem., 237, 1454-1459 (1962).
- Cases, S., Smith, S.J., Zheng, Y.W., Myers, H. M., Lear, S.R., Sande, E., Novak, S., Collins, C., Welch, C.B., Lusis, A.J., Erickson, S.K. and Farese, R.V., Jr. Identification of a gene encoding an acyl CoA:diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc. Natl. Acad. Sci. U.S.A., 95, 13018-13023 (1998) (DOI: 10.1073/pnas.95.22.13018).
- Cases, S., Stone, S.J., Zhou, P., Yen, E., Tow, B., Lardizabal, K.D., Voelker, T. and Farese, R.V., Jr. Cloning of DGAT2, a second mammalian diacylglycerol acyltransferase, and related family members. J. Biol. Chem., 276, 38870–38876 (2001) (DOI: 10.1074/jbc.M106219200).
- McFie, P.J., Stone, S.L., Banman, S.L. and Stone, S.J. Topological orientation of acyl-CoA:diacylglycerol acyltransferase-1 (DGAT1) and identification of a putative active site histidine and the role of the N terminus in dimer/tetramer formation. J. Biol. Chem., 285, 37377-37387 (2010) (DOI: 10.1074/jbc.M110.163691).
- Liang, J.J., Oelkers, P., Guo, C., Chu, P., Dixon, J.L., Ginsberg, H.N. and Sturley, S.L. Overexpression of human diacylglycerol acyltransferase 1, acyl-CoA:cholesterol acyltransferase 1, or acyl-CoA:cholesterol acyltransferase 2 stimulates secretion of apolipoprotein B-containing lipoproteins in McA-RH7777 cells.. J. Biol. Chem., 279, 44938-44944 (2004) (DOI: 10.1074/jbc.M408507200).
- Yamazaki, T., Sasaki, E., Kakinuma, C., Yano, T., Miura, S. and Ezaki, O. Increased very low density lipoprotein secretion and gonadal fat mass in mice overexpressing liver DGAT1. J. Biol. Chem., 280, 21506-21514 (2005) (DOI: 10.1074/jbc.M412989200).
- King, A.J., Segreti, J.A., Larson, K.J., Souers, A.J., Kym, P.R., Reilly, R.M., Zhao, G., Mittelstadt, S.W. and Cox, B.F. Diacylglycerol acyltransferase 1 (DGAT-1) inhibition lowers serum triglycerides in the Zucker fatty rat and the hyperlipidemic hamster. J. Pharmacol. Exp. Ther., 330, 526-531 (2009) (DOI: 10.1124/jpet.109.154047).
- Cheng, D., Meegalla, R.L., He, B., Cromley, D.A., Billheimer, J.T. and Young, P.R. Human acyl-CoA:diacylglycerol acyltransferase is a tetrameric protein. Biochem. J., 359, 707–714 (2001).
- Yen, C.L., Stone, S.J., Koliwad, S., Harris, C. and Farese, R.V., Jr. Thematic Review Series: Glycerolipids. DGAT enzymes and triacylglycerol biosynthesis. J. Lipid Res., 49, 2283-2301 (2008) (DOI: 10.1194/jlr.R800018-JLR200)
- Shih, M.Y., Kane, M.A., Zhou, P., Yen, C.L., Streeper, R.S., Napoli, J.L. and Farese, R.V., Jr. Retinol esterification by DGAT1 is essential for retinoid homeostasis in murine skin. J. Biol. Chem., 284, 4292-4299 (2009) (DOI: 10.1074/jbc.M807503200).
- Lardizabal, K.D., Mai, J.T., Wagner, N.W., Wyrick, A., Voelker, T. and Hawkins, D.J. DGAT2 is a new diacylglycerol acyltransferase gene family. Purification, cloning, and expression in insect cells of two polypeptides from Mortierella ramanniana with diacylglycerol acyltransferase activity. J. Biol. Chem., 276, 38862–38869 (2001) (DOI: 10.1074/jbc.M106168200).
- Kuerschner, L., Moessinger, C. and Thiele, C. Imaging of lipid biosynthesis: how a neutral lipid enters lipid droplets. Traffic, 9, 338-352 (2008) (DOI: 10.1111/j.1600-0854.2007.00689.x).
- Stone, S.J., Levin, M.C., Zhou, P., Han, J., Walther, T.C. and Farese, R.V., Jr. The endoplasmic reticulum enzyme DGAT2 is found in mitochondria-associated membranes and has a mitochondrial targeting signal that promotes its association with mitochondria. J. Biol. Chem., 284, 5352-5361 (2009) (DOI: 10.1074/jbc.M805768200).
- Stone, S.J., Levin, M.C. and Farese, R.V., Jr. Membrane topology and identification of key functional amino acid residues of murine acyl-CoA:diacylglycerol acyltransferase-2. J. Biol. Chem., 281, 40273-40282 (2006) (DOI: 10.1074/jbc.M607986200).
- Stone, S.J., Myers, H.M., Watkins, S.M., Brown, B.E., Feingold, K.R., Elias, P.M. and Farese, R.V., Jr. Lipopenia and skin barrier abnormalities in DGAT2-deficient mice. J. Biol. Chem., 279, 11767-11776. (2004) (DOI: 10.1074/jbc.M311000200).
- Meegalla, R.L., Billheimer, J.T. and Cheng, D. Concerted elevation of acyl-coenzyme A:diacylglycerol acyltransferase (DGAT) activity through independent stimulation of mRNA expression of DGAT1 and DGAT2 by carbohydrate and insulin. Biochem. Biophys. Res. Commun., 298, 317–323 (2002) (DOI: 10.1016/S0006-291X(02)02466-X).
- Suzuki, R., Tobe, K., Aoyama, M., Sakamoto, K., Ohsugi, M., Kamei, N., Nemoto, S., Inoue, A., Ito, Y., Uchida, S., Hara, K., Yamauchi, T., Kubota, N., Terauchi, Y. and Kadowaki, T. Expression of DGAT2 in white adipose tissue is regulated by central leptin action. J. Biol. Chem., 280, 3331-3337 (2005) (DOI: 10.1074/jbc.M410955200).
- Lau, T.E. and Rodriguez, M.A. A protein tyrosine kinase associated with the ATP-dependent inactivation of adipose diacylglycerol acyltransferase. Lipids, 31, 277–283 (1996).
- Yu, Y.H., Zhang, Y., Oelkers, P., Sturley, S.L., Rader, D.J. and Ginsberg, H.N. Posttranscriptional control of the expression and function of diacylglycerol acyltransferase-1 in mouse adipocytes. J. Biol. Chem., 277, 50876–50884 (2002) (DOI: 10.1074/jbc.M207353200).
- Smith, S.J., Cases, S., Jensen, D.R., Chen, H.C., Sande, E., Tow, B., Sanan, D.A., Raber, J., Eckel, R.H. and Farese, R.V., Jr. Obesity resistance and multiple mechanisms of triglyceride synthesis in mice lacking DGAT. Nat. Genet., 25, 87–90 (2000) (DOI: 10.1038/75651).
In This Section
- Glycerophosphate and Acylglycerophosphate Acyltransferases
- Mammalian Diacylglycerol Acyltransferases (DGAT)
- Fatty Acid beta-Oxidation
- Regulation of Lipins and Their Role in Lipid Metabolism
- Phospholipid Biosynthesis
- Phospholipases
- Acylglycerol Lipases (Neutral Lipid Hydrolysis)
- Metabolism and Function of Very-Long-Chain Polyunsaturated Fatty Acids (>C24) in Mammals